Publications

.
Scientific articles
Papers published in peer reviewed journals and book chapters
2026
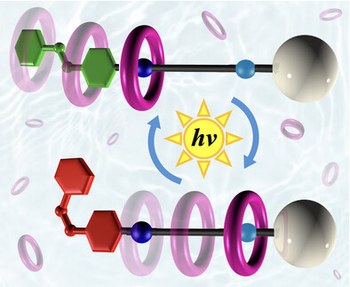
An artificial molecular pump powered by light
Federico Nicoli, Chiara Taticchi, Stefano Corra, Marina Tranfić Bakić, Massimo Baroncini, Serena Silvi, Jessica Groppi, Alberto Credi
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 16 April 2026
2026, e8103353
DOI: 10.1002/anie.8103353
Replicating the ability of biological systems to convert energy into directional molecular motion to perform functions is a central challenge in nanoscience. Artificial molecular pumps that can move substrates energetically uphill remain elusive, particularly when powered by light in an autonomous fashion. We report a molecular pump that uses light to actively transfer macrocycles from solution into a high-energy intramolecular compartment. The system operates via a photon-driven energy ratchet mechanism, sustaining a non-equilibrium distribution of species under continuous irradiation. All relevant kinetic and thermodynamic parameters were determined, and a comprehensive mechanistic model was developed. This minimalistic and robust design establishes a foundation for fully synthetic light-controlled non-equilibrium systems with potential applications in adaptive materials and solar energy conversion.
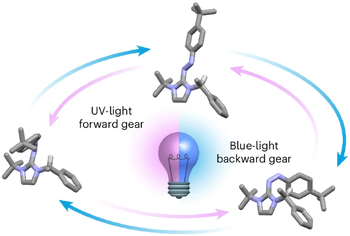
Wavelength-steered directional rotation in an autonomous light-driven molecular motor
Federico Nicoli, Chiara Taticchi, Emilio Lorini, Sara Borghi, Flavia Aleotti, Serena Silvi, Alberto Credi, Marco Garavelli, Luca Muccioli, Massimo Baroncini, Massimiliano Curcio
NATURE CHEMISTRY
Published online: 28 January 2026
2026, in press
DOI: 10.1038/s41557-025-02045-x
Artificial molecular motors are at the forefront of research in nanotechnology due to their ability to perform tasks by harnessing directionally controlled motion at the molecular scale. The development of light-driven nanomotors is a particularly challenging task that holds great potential for the development of sunlight-powered systems and active materials. Here we describe an azoimidazolium photochemical molecular rotary motor which operates along a triangular reaction cycle exploiting the formation of diastereomeric species upon photoisomerisation. The different thermal stability and photochemical reactivity of these diastereomers permit net directional motion combining a thermal rotation about a C–N single bond and two light-induced configurational rearrangements that proceed predominantly through a rotational mechanism, as corroborated by computational studies. The composition of the dissipative state obtained upon continuous supply of light can be modified by changing the irradiation wavelength, and as a result, the preferred rotation direction of the motor is inverted.
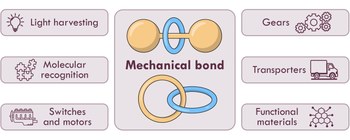
Molecular devices based on the mechanical bond: recent advances
Leonardo Andreoni, Chiara Taticchi, Brian Sachini, Alberto Credi
COORDINATION CHEMISTRY REVIEWS
Published online: 14 January 2026
2026, Vol. 553, art. n. 217583
DOI: 10.1016/j.ccr.2026.217583
The bottom-up construction and operation of nanoscale devices – namely, multicomponent (supra)molecular architectures capable of performing functions beyond the reach of their individual components – constitute a central focus in nanoscience and a compelling challenge in nanotechnology. Over the past three decades, the development of artificial molecular devices, including molecular machines and motors, has continuously stimulated the creativity of chemists, and the field continues to evolve at a remarkable pace. Among the strategies employed, the mechanical bond has emerged as a particularly powerful means of connecting molecular components to build functional systems. Mechanically interlocked molecules uniquely combine the robustness of covalent frameworks with the dynamic properties of non-covalent assemblies. Moreover, the precise modulation of steric and electronic interactions between mechanically bonded components offers rich opportunities for the emergence of novel functions. Recent advances in the design and synthesis of rotaxanes, catenanes and related interlocked structures have enabled the construction and operation of increasingly complex molecular devices and materials, many of which are capable of executing well-defined tasks. In this review, we present selected examples – classified according to their functional outputs – that exemplify the progress and potential of this rapidly developing area over the past decade.
2025
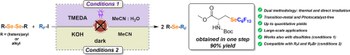
From light-driven to dark synthesis: streamlining perfluoroalkyl selenide ether formation
Loydel Torres Barroso, Romina S. Conde, Damian E. Yerien, Augustin A. Zottola, Al Postigo, Sebastian Barata-Vallejo
EUROPEAN JOURNAL OF ORGANIC CHEMISTRY
Published online: 2 November 2025
2025, art. n. e202500902
DOI: 10.1002/ejoc.202500902
Two complementary and operationally simple methods enable the efficient perfluoroalkylation of organodiselenides: (i) direct visible-light irradiation at 395 nm in aqueous MeCN with R(F)-I, requiring no photocatalyst, and (ii) a base-promoted protocol using KOH in MeCN under dark conditions. Both furnished perfluoroalkyl selenoethers in excellent yields, while only the photoinduced protocol delivered good to excellent yields of perfluoroalkyl thioethers from organodisulfides. The direct irradiation approach also enables the preparation of Se- and S-perfluoroalkylated derivatives of selenocysteine and cysteine, highlighting the applicability to biologically relevant substrates. Both protocols feature excellent chemical selectivity, good functional group tolerance, mild conditions, and simple operation. Compared with existing photocatalytic and thermal methods, these approaches provide greater versatility and operational simplicity, exemplified by the gram-scale synthesis of a perfluoroalkyl selenoether in excellent yield.
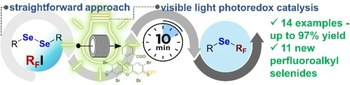
Visible-light-promoted R(F)-Se bond formation under continuous-flow. Synthesis of perfluoroalkyl selenide compounds
Juan Carlos Mundo-Zuna, Adrian A. Heredia, Lydia M. Bouchet, Al Postigo, Sebastian Barata-Vallejo, Juan E. Argüello, Gabriela Oksdath-Mansilla
JOURNAL OF FLOW CHEMISTRY
Published online: 13 October 2025
2025, Vol. 15, pp. 249-260
DOI: 10.1007/s41981-025-00364-1
The development of efficient and sustainable methodologies for the synthesis of fluoroalkyl chalcogen compounds has gained significant attention in recent years. In this study, we focus on the photoredox-catalyzed continuous-flow synthesis of perfluorinated chalcogenide compounds via the perfluoroalkylation of diaryl, dialkyl, dibenzyl, and diphenacyl diselenides with perfluoroalkyl iodides. The flow setup allowed superior irradiation efficiency, better mass and heat transfer, and reduced over-irradiation, leading to enhanced reaction performance compared to traditional batch methods. The methodology proved to be versatile across a wide range of substrates, including various organodiselenide compounds and perfluoroalkyl iodides [R(F)-I], demonstrating the influence of steric and electronic effects on reactivity. This research highlights the potential of continuous-flow technology for efficient, scalable photoredox catalysis, providing a versatile platform for the synthesis of perfluorinated compounds showcasing the effect of flow chemistry on reaction optimization and process intensification. Evidence for an electron transfer reaction involving TMEDA: R(F)-I complex is provided by cyclic voltammetry.
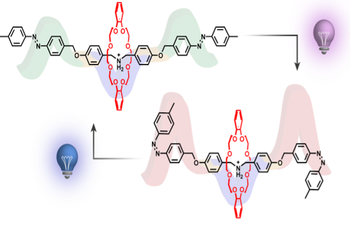
Light-driven molecular pumps: entanglement of thermodynamic and kinetic effects in the photocontrolled threading-unthreading of pseudorotaxanes
Brian Sachini, Chiara Taticchi, Massimo Baroncini, Stefano Corra, Alberto Credi
ORGANIC & BIOMOLECULAR CHEMISTRY
Published online: 26 September 2025
2025, Vol. 23, pp. 9171-9177
DOI: 10.1039/d5ob01154j
Light-powered molecular pumps represent an intriguing class of artificial nanomachines capable of using the energy of photons to perform directional transport. Pseudorotaxanes composed of macrocyclic crown ethers that encircle axles based on azobenzene photoswitches and secondary ammonium recognition sites have emerged as promising architectures, as light can modulate both the kinetics and thermodynamics of complex formation, thereby enabling directionally biased motion by an energy ratchet mechanism. In this study, we examine the effect of photoisomerization on the threading-unthreading dynamics of a symmetrical axle bearing decoupled azobenzene and dibenzylammonium units. The results are compared with those obtained on a previously reported more compact axle in which the two units share a phenyl ring. We found that, while Z-azobenzene significantly slows down the (un)threading kinetics with respect to the E-isomer, it does not destabilize the pseudorotaxane. Hence, such a decoupling challenges a core design requirement for photoinduced molecular pumps – namely, the light-induced modulation of both energy barriers and binding affinities. Our results underscore the critical role of electronic and spatial proximity between the photoisomerizable unit and the ring recognition site in achieving coupled kinetic and thermodynamic control. These insights provide refined design principles for the development of efficient light-driven molecular pumps based on modular supramolecular motifs.
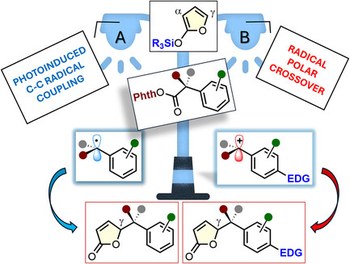
Harnessing vinylogy with radicals: photoinduced γ-benzylation reactions of 2-silyloxyfurans
Enrico Marcantonio, Debora Guazzetti, Luca Aimi, Kelly Bugatti, Pedro Mena, Marco Giannetto, Simone Fortunati, Andrea Sartori, Lucia Battistini, Leonardo Andreoni, Marco Lombardo, Franca Zanardi, Claudio Curti
ADVANCED SYNTHESIS & CATALYSIS
Published online: 16 July 2025
2025, Vol. 367, art. n. e70035
DOI: 10.1002/adsc.70035
2-Silyloxyfurans are among the most exploited Mukaiyama-type vinylogous nucleophiles from which a myriad of bioactive γ-butyrolactones have been accessed. Although a plethora of “polar” reactions featuring this scaffold as a key player have been developed so far, its behavior in radical chemistry is still in its infancy. Herein, the development of two complementary vinylogous, radical-mediated benzylations of 2-silyloxyfurans, promoted by visible light and suitable photoredox catalysts, is described. Common to both photocatalytic cycles is the reduction of a suitable redox active ester forging a key benzyl radical intermediate, which undergoes two different fates. First, the photoinduced oxidation of the silyloxyfuran generates an unprecedented silyl-radical cation species able to trap the nucleophilic benzyl radical. Alternatively, with specific electron-rich substrates, the benzyl radical can be converted to the corresponding benzyl carbocation intermediate via a net-neutral radical-polar crossover pathway, enabling a vinylogous, polar benzylation reaction. A broad scope of chiral, γ-benzyl butenolides is obtained in one step, some of which are used as strategic precursors to access bioactive phenyl-γ-valerolactone metabolites.
Defying chemical equilibrium with light
Brian Sachini, Stefano Corra, Massimiliano Curcio, Jessica Groppi, Alberto Credi
TRENDS IN CHEMISTRY
Published online: 20 May 2025
2025, Vol. 7, pp. 372-383
DOI: 10.1016/j.trechm.2025.04.009
Chemical systems capable of exploiting an energy source to function away from thermodynamic equilibrium are a hallmark of the biological world. The design and operation of artificial versions of such systems constitute a stimulating and highly challenging scientific goal with considerable expectations for application in synthetic chemistry, catalysis, materials science, energy conversion, and medical therapy. In this context, supplying energy in the form of photons has several elements of conceptual interest and practical advantage. Here we present the progress of research over the past 5 years on artificial molecular-based systems that can use light energy to operate away from equilibrium – an emerging field that has just begun to express its innovative potential.
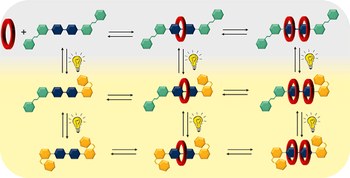
An azobenzene-bipyridinium derivative as a component in the construction of photoresponsive pseudorotaxanes
Leonardo Andreoni, Dalila Cafagno, Alberto Credi, Jessica Groppi
SYNLETT
Published online: 7 May 2025
2025, Vol. 36, pp. 1574-1578
DOI: 10.1055/s-0043-1775482
We describe the synthesis and characterization of a bipyridinium derivative conjugated with two azobenzene groups (Azo2Bpy2+). The design maintains the ability of the photoswitches to undergo E to Z isomerization upon irradiation to generate a mixture of EE, EZ and ZZ isomers. Moreover, Azo2Bpy2+ is able to undergo self-assembly with dibenzo[24]crown-8 ether to generate [3]-pseudorotaxanes, driven by the strong cooperative effects, for all its geometric isomers.
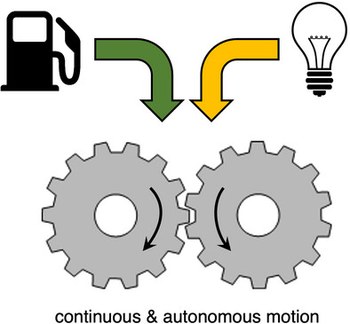
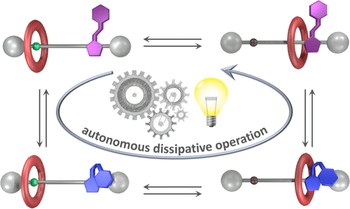
Autonomous artificial molecular motors and pumps
Chiara Taticchi, Massimiliano Curcio, Stefano Corra
CHEMSYSTEMSCHEM
Published online: 28 April 2025
2025, Vol. 7, e202400101
DOI: 10.1002/syst.202400101
Over the past decade there has been a tremendous development of systems capable to autonomously convert energy, in particular light and chemical, into directed motion at the nanoscale. These nanoscopic devices are called molecular motors. The autonomous operation of artificial molecular motors and pumps under constant experimental conditions represents a key achievement to their implementation into more sophisticated networks. Nonetheless, the principles behind successful autonomous operation are only recently being rationalized. Within this review we focus on the fundamental aspects that enable the autonomous operation of molecular motors exploiting light and chemical energy. We also compare the mechanisms of operation with these two energy sources and highlight the common ground of these systems as well as their differences and specificities by discussing a selection of recent examples in the two classes. Finally, we provide a perspective view on future advances in this exciting research area.

Back to fundamentals of research: ethics and academic freedom. Position paper.
Alberto Credi, Luca Fontanesi, Rebecca Montanari, Sara Coppini, Bianca Gualandi
AMS ACTA
Published online: 11 February 2025
DOI: 10.6092/unibo/amsacta/8225
On December 2, 2024, the University of Bologna organised an international event titled: “Back to Fundamentals of Research: Ethics and Academic Freedom.” The event featured prominent speakers and experts from national and international institutions, who shared their insights on emerging ethical concerns and the risks to academic freedom. Through this position paper, the University of Bologna aims to highlight the key messages that emerged from the discussions and to outline the challenges and recommendations to create a more responsible and supportive research environment.

Pseudomorphic transformation in nanostructured thiophene-based materials
Mattia Zangoli, Raffaello Mazzaro, Eugenio Lunedei, Eduardo Fabiano, Ilse Manet, Andrea Candini, Alessandro Kovtun, Meriem Goudjil, Alberto Zanelli, Shlomo Rozen, Massimo Gazzano, Massimo Baroncini, Francesca Di Maria
ACS NANO
Published online: 8 January 2025
2025, Vol. 19, pp. 2245-4460
DOI: 10.1021/acsnano.4c11681
This study reveals the capability of nanostructured organic materials to undergo pseudomorphic transformations, a ubiquitous phenomenon occurring in the mineral kingdom that involves the replacement of a mineral phase with a new one while retaining the original shape and volume. Specifically, it is demonstrated that the postoxidation process induced by HOFCH3CN on preformed thiophene-based 1D nanostructures preserves their macro/microscopic morphology while remarkably altering their electro-optical properties by forming a new oxygenated phase. Experimental evidence proves that this transformation proceeds via an interface-coupled dissolution-precipitation mechanism, leading to the growth of a porous oxidized shell that varies in thickness with exposure time, enveloping the pristine smooth core. The oxygenated species exhibits stronger electron-acceptor characteristics than the core material, promoting charge transfer state formation, as confirmed by microspectroscopy and DFT calculations. This enables (i) precise modulation of the nanostructure's surface potential, allowing for the formation of entirely organic heterojunctions with precise spatial resolution via wet chemical processing; (ii) effective doping of the nanostructure, resulting in a strong change of the conductivity temperature dependence and a switch between a low and high conduction state depending on the applied bias. Overall, this work showcases an approach to engineering "impossible" composite architectures with pre-established morphology and tailored chemical-physical properties.

Light-driven ratcheted synthesis of diastereomeric host-guest systems
Iago Neira, Chiara Taticchi, Federico Nicoli, Massimiliano Curcio, Marcos D. García, Carlos Peinador, Serena Silvi, Massimo Baroncini, Alberto Credi
CHEM
Published online: 27 December 2024
2025, Vol. 11, 102375
DOI: 10.1016/j.chempr.2024.11.013
The ability to exploit an energy source to drive chemical reactions away from thermodynamic equilibrium is an essential feature of life and a grand challenge for the design of fuel-driven dynamic artificial nanosystems. Here, we investigate the effect of light irradiation on the formation of supramolecular complexes composed of azobenzene-type guests and a cyclodextrin (CD) host in water. Whereas previous studies on these complexes have focused on equilibrium properties, our work explores far-from-equilibrium distributions obtained by light-driven association. We demonstrate that the relative abundance of the two CD orientational diastereomeric complexes can be inverted upon photoirradiation and showcase a ratcheted approach, employing biocompatible macrocycles and harnessing visible light, to the spontaneous formation of high-energy CD complexes with broad applicability in aqueous environments. We foresee opportunities for the development of active materials, the design of artificial metabolic networks, and the engineering of molecular machines operating under physiological conditions.
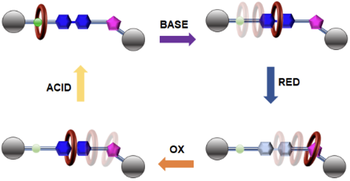
Directional ring translation in a pH- and redox-driven tristable [2]rotaxane
Leonardo Andreoni, Jessica Groppi, Özlem Seven, Massimo Baroncini, Alberto Credi, Serena Silvi
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 20 September 2024
2025, Vol. 64, e202414609
DOI: 10.1002/anie.202414609
We describe the synthesis and characterization of a [2]rotaxane comprising a dibenzo-24-crown-8 (DB24C8) macrocyclic component and a thread containing three recognition sites: ammonium (AmH+), bipyridinium (Bpy2+) and triazolium (Trz+). AmH+ and Bpy2+ are responsive to fully orthogonal stimuli, pH and electrochemical, which allows to precisely control the directional translation of the macrocycle along the axle. A better understanding of the processes driving the operation of the system was obtained thanks to an in-depth thermodynamic characterization. Orthogonal stimuli responsive tristable rotaxanes represent the starting point for the creation of linear motors and the development of molecular logic gates.
2024

Back to fundamentals of research: interdisciplinarity. Position paper
Alberto Credi, Anna Chiara Fariselli, Rebecca Montanari, Sara Coppini, Alberto Furgoni, Bianca Gualandi, Letizia Marongiu
AMS ACTA
Published online: 13 December 2024
DOI: 10.6092/unibo/amsacta/8074
On October 9, 2024, the University of Bologna held an international event titled: “Back to fundamentals of research: Interdisciplinarity”. The event featured distinguished speakers and experts from national and international institutions who shared their insights on interdisciplinary research and recommendations on how to foster this approach. Through this position paper, the University of Bologna intends to highlight the key messages that emerged from the discussions and point out the challenges and recommendations to create a more supportive environment for interdisciplinary research.
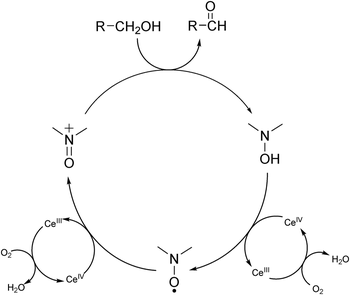
Rotaxane-catalyzed aerobic oxidation of primary alcohols
Ilario Baù, Cecilia Poderi, Francesca Sardu, Alessia Giancola, Anna Turchetti, Paola Franchi, Lorenzo Casimiro, Leonardo Andreoni, Serena Silvi, Elisabetta Mezzina, Marco Lucarini
COMMUNICATIONS CHEMISTRY
Published online: 27 November 2024
2024, Vol. 7, art. n. 278
DOI: 10.1038/s42004-024-01375-0
Nitroxide radicals are widely utilized as catalysts for the oxidation of primary alcohols. Here, the aerobic catalytic oxidation cycle of nitroxide radicals has been implemented within a mechanically interlocked rotaxane architecture consisting of a paramagnetic crown ether, which is confined by a molecular axle containing a dialkylammonium station and a 1,2,3-triazole unit. The rotaxane is engineered to exploit the oxidation of a primary alcohol: the primary catalyst is the wheel, a nitroxide radical capable of altering its oxidation state during the catalytic cycle, while the co-oxidant is the Cerium(IV)/O2 couple. The synthesis of the proposed rotaxane, along with its characterization using EPR, HRMS, voltammetry and NMR data, is reported in the paper. The aerobic catalytic oxidation cycle was further investigated using EPR, NMR and GC-MS analyses. This study can aid in the design of autonomously driven molecular machines that exploit the aerobic catalytic oxidation of nitroxide radicals.

Il ruolo dello scienziato nel mondo di oggi
Alberto Credi
ANNALES. PROCEEDINGS OF THE ACADEMY OF SCIENCES OF BOLOGNA. CLASS OF PHYSICAL SCIENCES
Published online: 12 November 2024
2024, Vol. 2, pp. 151-157
DOI: 10.30682/annalesps2402l
Scientific research underpins socioeconomic progress and will enable mankind to address the major challenges of our time through currently unknown solutions. In particular, curiosity-driven fundamental research is essential to achieve groundbreaking innovation. To reach this goal, scientists must focus on the quality of their work and uphold the core values of research: integrity, ethics, transparency, reproducibility, collaboration across geographic boundaries, and interdisciplinary dialogue. Passion, creativity, humility and perseverance are essential qualities of the scientist, along with the ability to communicate effectively with the public and policymakers, to generate trust for research and support informed decisions at all levels.
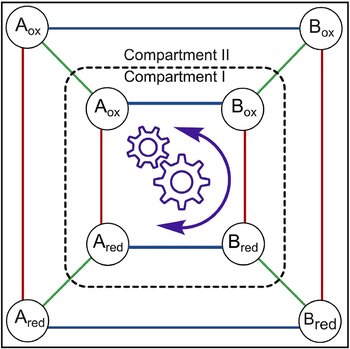
Analysis of kinetic asymmetry in a multi-cycle reaction network establishes the principles for autonomous compartmentalized molecular ratchets
Emanuele Penocchio, Ahmad Bachir, Alberto Credi, R. Dean Astumian, Giulio Ragazzon
CHEM
Published online: 13 September 2024
2024, Vol. 10, pp. 3644-3655
DOI: 10.1016/j.chempr.2024.07.038
Kinetic asymmetry is a key parameter describing non-equilibrium systems: it indicates the directionality of a reaction network under steady-state conditions. So far, kinetic asymmetry has been evaluated only in networks featuring a single cycle. Here, we have investigated kinetic asymmetry in a multi-cycle system using a combined theoretical and numerical approach. First,we report the general expression of kinetic asymmetry for multi-cycle networks. Then, we specify it for a recently reported electrochemically controlled network comprising diffusion steps, which we used as a model system to reveal how key parameters influence directionality. In contrast with the current understanding,we establish that spatial separation - including compartmentalization - can enable autonomous energy ratchet mechanisms, with directionality dictated by thermodynamic features. Kinetic simulations confirm analytical findings and illustrate the interplay between diffusion, chemical, and electrochemical processes. The treatment is general, as it can be applied to other multi-cycle networks, facilitating the realization of endergonic processes across domains.

Back to fundamentals of research: reliability, transparency and reproducibility. Position paper
Alberto Credi, Francesca Masini, Stefano Diciotti, Monica Forni, Silvio Peroni, Sara Coppini, Bianca Gualandi
AMS ACTA
Published online: 24 July 2024
DOI: 10.6092/unibo/amsacta/7803
On May 20, 2024, the University of Bologna held an international event titled: “Back to fundamentals of research: Reliability, Transparency and Reproducibility” with distinguished experts from national and international institutions. The debate revolved around what it means to perform research responsibly and whether the reproducibility of results can be used as a reliable proxy for research quality.
Through this position paper, the University of Bologna intends to highlight some key messages that emerged from the discussions and point out challenges and recommendations in ensuring reproducibility and trustworthy methodologies in research environments.
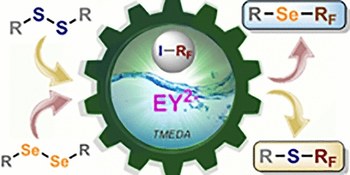
Photocatalytic perfluoroalkylation of disulfides and diselenides. Syntheses of perfluoroalkyl thio- and seleno-ethers
Romina S. Conde, Loydel T. Barroso, Sheila G. Perez Edighill, Damian E. Yerien, Beatriz Lantaño, Massimo Baroncini, Sebastian Barata-Vallejo, Al Postigo
JOURNAL OF ORGANIC CHEMISTRY
Published online: 21 July 2024
2024, Vol. 89, pp. 10867-10877
DOI: 10.1021/acs.joc.4c01149
The synthesis of alkyl(aryl)-fluoroalkyl sulfanyl [R(Ar)-S-R-F] and aryl-fluoroalkyl selenolyl (Ar-Se-R-F) ethers through visible-light photocatalysis has been successfully carried out. This process involves disulfides, and diselenides [R(Ar)-X-X-R(Ar), where X = S or Se], and fluoroalkyl iodides (R-F-I) in the presence of a base as an additive under photocatalysis. The photocatalyst Eosin Y and green light-emitting diodes are utilized for irradiation of R(Ar)-S-R-F and Ar-Se-R-F syntheses. Our method integrates low-energy visible-light photocatalysis, commercially available perfluoroalkylating reagents (R-F-I), and easily obtainable disulfides and diselenides as starting materials. Mechanistic studies and a detailed synthetic procedure for (Ar)-S-R-F on a large scale are presented.
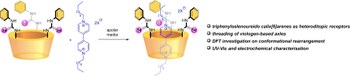
Selenoureido calix[6]arenes: a novel platform for pseudorotaxane synthesis
Federica Cester Bonati, Leonardo Andreoni, Silvia Cattani, Caterina Baccini, Sara Anzellotti, Gianpiero Cera, Serena Silvi, Andrea Secchi
EUROPEAN JOURNAL OF ORGANIC CHEMISTRY
Published online: 23 May 2024
2024, Vol. 27, art. n. e202400237
DOI: 10.1002/ejoc.202400237
Heteroditopic calix[6]arenes are widely employed macrocycles for the synthesis of pseudo-oriented interlocked systems and stimuli-induced molecular machines, among others. Herein, we introduce a new calix[6]arene receptor functionalised with three phenylselenoureido groups. These hydrogen-bonding donor moieties are able to promote the threading of viologen-based organic axles inside the macrocyclic cavity to form a stable pseudorotaxane species. Preliminary investigations using 1H-NMR measurements and semi-empirical and DFT studies suggested its further use as a platform for the synthesis of rotaxanes.
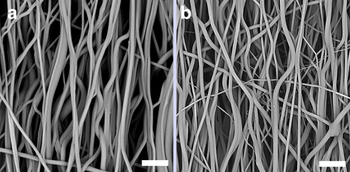
Electrochemically responsive nanofibers by stimulation of a dialkyl-bipyridinium molecular switch
Elisabetta Petri, Carlo Gotti, Jessica Groppi, Anna Liguori, Serena Silvi, Alberto Credi, Andrea Zucchelli, Francesca Soavi, Maria Letizia Focarete, Chiara Gualandi, Catia Arbizzani
MATERIALS CHEMISTRY AND PHYSICS
Published online: 3 March 2024
2024, Vol. 317, 129155
DOI: 10.1016/j.matchemphys.2024.129155
The translation of the well-known reversible electrochemical switching of viologen species from solution to the solid state may open enormous possibilities for developing electrically responsive materials. In this work, we present and discuss the electrochemical response of a viologen derivative, 1,1′-dipropyl-4,4′-bipyridinium bis-hexafluorophosphate (Bpy2+), embedded into a polyurethane fibrous matrix. The results showcase the possibility of stimulating electrochemically the active molecule inside the polymer fibers, both in random and aligned configurations, retaining the fibrous morphology and structure while avoiding the use of conductive polymers and ionic liquids, thus opening a new scenario in electrochemically responsive soft actuators.
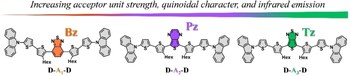
Multifunctional photoelectroactive materials for optoelectronic applications based on thieno[3,4-b]pyrazines and thieno[1,2,5]thiadiazoles
Mattia Zangoli, Filippo Monti, Alberto Zanelli, Martina Marinelli, Soraia Flammini, Nicol Spallacci, Anna Zakrzewska, Massimiliano Lanzi, Elisabetta Salatelli, Filippo Pierini, Francesca Di Maria
CHEMISTRY - A EUROPEAN JOURNAL
Published online: 20 November 2023
2024, Vol. 30, art. n. e202303590
DOI: 10.1002/chem.202303590
In this study, we introduce a novel family of symmetrical thiophene-based small molecules with a Donor-Acceptor-Donor structure. These compounds feature three different acceptor units: benzo[c][1,2,5]thiadiazole (Bz), thieno[3,4-b]pyrazine (Pz), and thieno[1,2,5]thiadiazole (Tz), coupled with electron donor units based on a carbazole-thiophene derivative. Using Density Functional Theory (DFT), we investigate how the molecular geometry and strength of the central acceptor unit impact the redox and spectroscopic properties. Notably, the incorporation of Pz and Tz moieties induces a significant redshift in the absorption and emission spectra, which extend into the near-infrared (NIR) region, simultaneously reducing their energy gaps (similar to 1.4-1.6 eV). This shift is attributed to the increased coplanarity of the oligomeric inner core, both in the ground (S0) and excited (S1) states, due to the enhanced quinoidal character as supported by bond-length alternation (BLA) analysis. These structural changes promote better pi-electron delocalization and facilitate photoinduced charge transfer processes in optoelectronic devices. Notably, we show that Pz- and Tz-containing molecules exhibit NIR electrochromic behavior and present ambivalent character in bulk heterojunction (BHJ) solar cells. Finally, theoretical calculations suggest that these molecules could serve as effective two-photon absorption (2PA) probes, further expanding their potential in optoelectronic applications.
2023
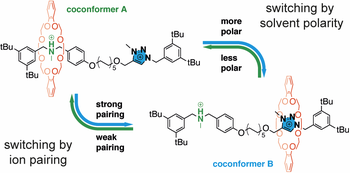
Controlling molecular shuttling in a rotaxane with weak ring recognition sites
Nina Bukhtiiarova, Stefano Corra, Alberto Credi
CHEMICAL COMMUNICATIONS
Published online: 9 October 2023
2023, Vol. 59, pp. 13159-13162
DOI: 10.1039/d3cc04483a
We describe a rotaxane molecular shuttle encompassing a triazolium and a tertiary ammonium units as weak recognition sites for the ring. Such a design, which differs from that of typical controllable rotaxanes, allows to precisely tune the ring distribution among the two sites – i.e. , the coconformational equilibrium – by changing the solvent polarity or the nature of the counteranions. Shuttling of the ring between the two stations can also be toggled by acid-base stimuli. Such an approach is paradigmatic to obtain rotaxanes that can sense environmental changes and transduce them into a coconformational response and opens avenues for novel applications in sensing and stimuli-responsive materials.
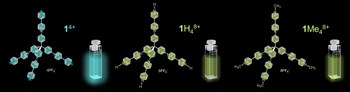
Elucidating the excited state behavior of pyridyl pyridinium systems via computational and transient absorption studies of tetrahedral multichromophoric arrays and their model compounds
Barbara Ventura, Daniele Veclani, Alessandro Venturini, Nicola Armaroli, Massimo Baroncini, Paola Ceroni, Marianna Marchini
CHEMISTRY - A EUROPEAN JOURNAL
Published online: 10 August 2023
2023, Vol. 29, art. n. e202301853
DOI: 10.1002/chem.202301853
The tetrahedral shape-persistent molecule 1(4+), containing four identical pyridyl pyridinium units connected via a sp3 hybridized carbon atom, has been investigated in detail by means of steady-state and time resolved spectroscopy. Remarkable photophysical properties are observed, particularly in comparison with protonated and methylated analogues [1H4(8+), 1Me4(8+)], which exhibit substantially shorter excited state lifetimes and lower emission quantum yields. Theoretical studies have rationalized the behavior of the tetrameric molecules relative to the monomers, with DFT and TD-DFT calculations corroborating steady-state (absorption and emission) and transient absorption spectra. The behavior of the monomeric compounds [each consisting in one of the four identical subunits of the tetramers, i. e., 2(+), 2H(2+) and 2Me(2+)] considerably differs from that of the tetramers, indicating a strong electronic interaction between the subunits in the tetrameric species, likely promoted by the homoconjugation through the connecting sp3 C atom. 2(+) is characterized by a peculiar S1-S2 excited state inversion, whereas the short-lived emitting S1 state of 2H(2+) and 2Me(2+) exhibits a partial charge-transfer character, as substantiated by spectro-electrochemical studies. Among the six investigated systems, only 1(4+) is a sizeable luminophore (Fem=0.15), which is related to the peculiar features of its singlet state.
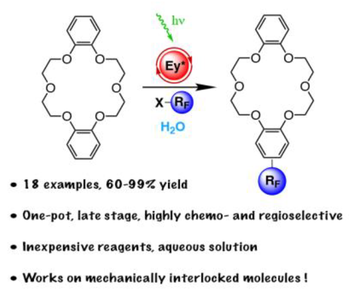
Late-stage photocatalytic fluoroalkylation of aromatic crown ethers in aqueous media
Damian E. Yerien, Jessica Groppi, Al Postigo, Alberto Credi, Romina S. Conde, Massimo Baroncini, Sebastian Barata-Vallejo
EUROPEAN JOURNAL OF ORGANIC CHEMISTRY
Published online: 26 May 2023
2023, Vol. 26, e202300478
DOI: 10.1002/ejoc.202300478
Modified crown ethers are fundamental building blocks in supramolecular chemistry, with applications in phase transfer catalysis, metal extraction, smart materials, and molecular machines. Here we report on a one-pot highly chemo- and regioselective photocatalytic fluoroalkylation protocol for the mono-functionalization of benzo substituted crown ethers. For this important class of macrocycles, the method described here represents one of the few late-stage functionalization procedures that is both high yielding and scalable. Because of its breadth of scope and substrate tolerance, the devised approach can be applied to a wide range of aromatic crown ethers (18 examples, up to 99% yield), including those engaged in mechanically interlocked molecules.

Photoactivated artificial molecular motors
Stefano Corra, Massimiliano Curcio, Alberto Credi
JACS AU
Published online: 8 May 2023
2023, Vol. 3, pp. 1301-1313
DOI: 10.1021/jacsau.3c00089
Accurate control of long-range motion at the molecular scale holds great potential for the development of ground-breaking applications in energy storage and bionanotechnology. The past decade has seen tremendous development in this area, with a focus on the directional operation away from thermal equilibrium, giving rise to tailored man-made molecular motors. As light is a highly tunable, controllable, clean, and renewable source of energy, photochemical processes are appealing to activate molecular motors. Nonetheless, the successful operation of molecular motors fueled by light is a highly challenging task, which requires a judicious coupling of thermal and photoinduced reactions. In this paper, we focus on the key aspects of light-driven artificial molecular motors with the aid of recent examples. A critical assessment of the criteria for the design, operation, and technological potential of such systems is provided, along with a perspective view on future advances in this exciting research area.
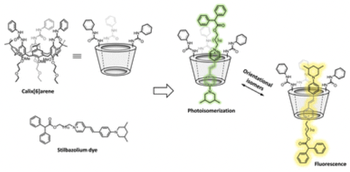
Selective enhancement of organic dye properties through encapsulation in rotaxane orientational isomers
Leonardo Andreoni, Federica Cester Bonati, Jessica Groppi, Davide Balestri, Gianpiero Cera, Alberto Credi, Andrea Secchi, Serena Silvi
CHEMICAL COMMUNICATIONS
Published online: 28 March 2023
2023, Vol. 59, pp. 4970-4973
DOI: 10.1039/d3cc01135f
We report the synthesis and characterisation of [2]rotaxanes based on a stilbazolium dye and a calix[6]arene macrocycle. Since both components are non-symmetric, two orientational isomers are obtained. The two isomers display distinct photophysical and photochemical properties in solution and solid state, superior to the unencapsulated dye.
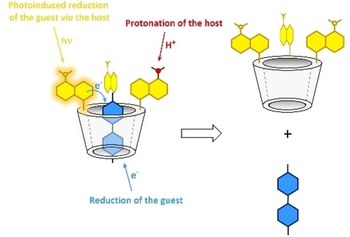
A multiresponsive calix[6]arene pseudorotaxane empowered by fluorophoric dansyl groups
Leonardo Andreoni, Giovanni Mariano Beneventi, Gabriele Giovanardi, Gianpiero Cera, Alberto Credi, Arturo Arduini, Andrea Secchi, Serena Silvi
CHEMISTRY - A EUROPEAN JOURNAL
Published online: 16 March 2023
2023, Vol. 29, e202203472
DOI: 10.1002/chem.202203472
A multiresponsive pseudorotaxane, composed by a dansyl calix[6]arene and a bipyridinium axle, is presented. The disassembly of this complex can be triggered by: i) protonation of the dansyl units (chemical stimulus); ii) electrochemical reduction of the bipyridinium unit (electrochemical stimulus); iii) photochemical reduction of the bipyridinium unit, enabled by photoinduced electron transfer from the dansyl units (light stimulus). Overall, three orthogonal and reversible stimuli can be employed to control the stability of this novel pseudorotaxane.

Photochemically driven molecular machines based on coordination compounds
Alberto Credi, Serena Silvi, Massimo Baroncini, Leonardo Andreoni, Chiara Taticchi
in COMPREHENSIVE INORGANIC CHEMISTRY III (3rd Ed.)
J. Reedijk , K. Poeppelmeier (Eds.), Elsevier, Waltham (USA)
Published online: 1 March 2023
2023, Vol. 8, Ch. 10, pp. 417-438
DOI: 10.1016/B978-0-12-823144-9.00094-7
The marriage of molecular machines and coordination compounds has been fruitful since the early stages of the research on molecular machines. First only structural elements, but soon also functional components, metal complexes played a crucial part in the development of sophisticated systems, thanks to their excellent and versatile geometrical, photophysical and electrochemical properties. In this Chapter photoactive molecular machines will be divided in three main classes, depending on the role of the coordination compounds: structural elements, triggers of the photochemical processes, or photoactive structural components.
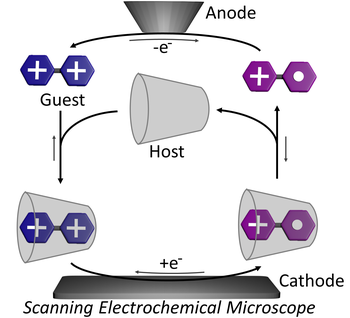
Autonomous non-equilibrium self-assembly and molecular movements powered by electrical energy
Giulio Ragazzon, Marco Malferrari, Arturo Arduini, Andrea Secchi, Stefania Rapino, Serena Silvi, Alberto Credi
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 24 November 2022
2023, Vol. 62, e202214265
DOI: 10.1002/anie.202214265
The ability to exploit energy autonomously is one of the hallmarks of Life. Mastering such processes in artificial nanosystems can open technological opportunities. In the last decades, light- and chemically-driven autonomous systems have been developed in relation to conformational motion and self-assembly, mostly in relation to molecular motors. On the contrary, despite electrical energy is an attractive energy source to power nanosystems, its autonomous harnessing received little attention. Herein we consider an operation mode allowing the autonomous exploitation of electrical energy by a self-assembling system. Threading and dethreading motions of a pseudorotaxane take place autonomously in solution, powered by the current flowing between the electrodes of a scanning electrochemical microscope. The underlying autonomous energy ratchet mechanism drives the self-assembly steps away from equilibrium with a higher energy efficiency compared to other autonomous systems. The strategy is general and might be extended to other redox-driven systems.
2022
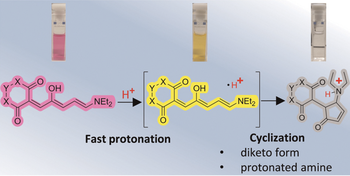
Acidochromism of donor-acceptor Stenhouse adducts in organic solvent
Antonio Fiorentino, Brian Sachini, Stefano Corra, Alberto Credi, Cristina Femoni, Aurore Fraix, Serena Silvi
CHEMICAL COMMUNICATIONS
Published online: 15 August 2022
2022, Vol. 58, pp. 11236-11239
DOI: 10.1039/d2cc03761k
First generation DASA derivatives can be reversibly isomerized from the coloured, open form to the colourless, closed isomer upon protonation, thus behaving as acidochromic compounds in halogenated organic solvent

Light-driven molecular machines
Alberto Credi, Serena Silvi, Massimo Baroncini
in MOLECULAR PHOTOSWITCHES-CHEMISTRY, PROPERTIES, AND APPLICATIONS
Z. L. Pianowski (Ed.), Wiley-VCH, Weinheim, Germany
Published online: 3 June 2022
2022, Ch. 32, pp. 735-784
DOI: 10.1002/9783527827626.ch32
In the last 40 years, the opinion of most researchers has moved from considering artificial molecular machines as simple laboratory curiosities to viewing them as valuable tools to impart novel functionalities and properties to advanced materials and multicomponent systems. In this chapter, we present an overview of the progress on the design and realization of light-activated molecular machines and on their different applications. This research field has always been in close relationship with the study of photoswitchable and photochromic compounds. Indeed, it can be confidently said that at the heart of any light-activated nanomachine, a photoswitchable moiety is at work.
After a brief description of the basic type of photoinduced processes that light-activated nanomachines exploit for their operation, a concise description of the main classes of nanomachines is presented. The chapter follows with examples in which the nanoscale motion of the machine is translated in an advanced functionality. These include switchable catalysts, linear and rotary transporters of small molecular cargos, and supramolecular pumps. Furthermore, examples in which the collective operation of an assembly of molecular machines is harnessed to perform tasks at the microscale and macroscale in hard and soft materials are also reviewed. In particular, functionalized surfaces, metal–organic frameworks, polymers, and self-assembled nanostructures are described wherein the nanoscale movement of embedded molecular machines is amplified, allowing the realization of muscle-like actuators, microfluidic devices, and polymeric materials for light energy transduction and storage.
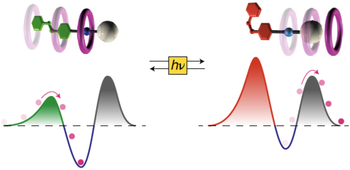
Kinetic and energetic insights into the dissipative non-equilibrium operation of an autonomous light-powered supramolecular pump
Stefano Corra, Marina Tranfić Bakić, Jessica Groppi, Massimo Baroncini, Serena Silvi, Emanuele Penocchio, Massimiliano Esposito, Alberto Credi
NATURE NANOTECHNOLOGY
Published online: 27 June 2022
2022, Vol. 17, pp. 746-751
DOI: 10.1038/s41565-022-01151-y
Natural and artificial autonomous molecular machines operate by constantly dissipating energy coming from an external source to maintain a non-equilibrium state. Quantitative thermodynamic characterization of these dissipative states is highly challenging as they exist only as long as energy is provided. Here we report on the detailed physicochemical characterization of the dissipative operation of a supramolecular pump. The pump transduces light energy into chemical energy by bringing self-assembly reactions to non-equilibrium steady states. The composition of the system under light irradiation was followed in real time by 1H NMR for four different irradiation intensities. The experimental composition and photon flow were then fed into a theoretical model describing the non-equilibrium dissipation and the energy storage at the steady state. We quantitatively probed the relationship between the light energy input and the deviation of the dissipative state from thermodynamic equilibrium in this artificial system. Our results provide a testing ground for newly developed theoretical models for photoactivated artificial molecular machines operating away from thermodynamic equilibrium.
Mechanically interlocked systems: photoactive rotaxanes and catenanes
Massimo Baroncini, Martina Canton, Lorenzo Casimiro, Alberto Credi, Serena Silvi
in SPRINGER HANDBOOK OF INORGANIC PHOTOCHEMISTRY
D. Bahnemann, A. O. T. Patrocinio (Eds.), Springer Nature, Cham, Switzerland
Published online: 26 June 2022
2022, Ch. 22, pp. 585-602
DOI: 10.1007/978-3-030-63713-2_22
The mechanical bond offers novel and intriguing opportunities to connect together molecular components and arrange them in space. Mechanically interlocked molecules(MIMs) such as rotaxanes and catenanes can indeed be designed to operate as molecular devices, that is, to accomplish function(s) that arise(s) from the cooperation of their molecular components. In this chapter we will deal with rotaxane- and catenane-based architectures characterized by two main features: (i) the presence of inorganic moieties in the molecular structure and (ii) the integration
of photoactive units. Here we focus on metal complexes as inorganic moieties, which can play the dual role of scaffolds for the construction of the molecules and for controlling the spatial arrangement of the components, and of functional units, because they present peculiar photophysical
and electrochemical properties. The use of light to operate molecular devices and machines has long been acknowledged as a most valuable choice under several aspects. In this regard, for the sake of clarity, we have classified the selected examples in two main categories: photoactive systems, which are characterized by photoinduced processes within the components of the interlocked architecture, and photoactivated systems, wherein light is used to cause a mechanical rearrangement of the components. The examples discussed will show how the union of the structural control offered by the mechanical bond with the tools of inorganic chemistry can lead to the realization of complex structures with sophisticated functions.

Multimodal sensing in rewritable, datamatrix azobenzenes based devices
Alex Boschi, Simone Cinili, Eva Bystrenova, Giampiero Ruani, Massimo Baroncini, Alberto Credi, Andrea Candini, Denis Gentili, Massimiliano Cavallini
JOURNAL OF MATERIALS CHEMISTRY C
Published online: 22 June 2022
2022, Vol. 10, pp. 10132-10138
DOI: 10.1039/D2TC01565J
Here, we exploited the trans-cis photoisomerization switching of an azobenzenes compound for multimodal sensing to UV light and to fabricate multimodal readable and rewritable, datamatrix azobenzenes based devices. We first demonstrated that sensing capabilities to UV light can be simultaneously monitored by the change of optical, spectroscopic and electrical properties. Then we exploited this capability by integrating tetra(azobenzene)methane crystals in a micrometric TAG whose information can be modified and repristinated by local UV treatment and thermal annealing. The system was characterised by polarized optical microscopy, Raman spectroscopy, conductive atomic force microscopy and Kelvin Probe Force Microscopy.
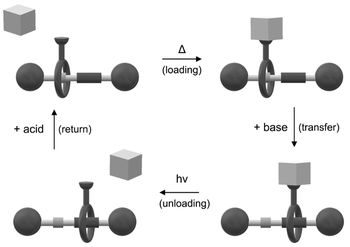
Photoinduced autonomous nonequilibrium operation of a molecular shuttle by combined isomerization and proton transfer through a catalytic pathway
Federico Nicoli, Massimiliano Curcio, Marina Tranfić Bakić, Erica Paltrinieri, Serena Silvi, Massimo Baroncini, Alberto Credi
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY
Published online: 16 May 2022
2022, Vol. 144, pp. 10180-10185
DOI: 10.1021/jacs.1c13537
We describe a [2]rotaxane whose recognition sites for the ring are a dibenzylammonium moiety, endowed with acidic and H-bonding donor properties, and an imidazolium center bearing a photoactive phenylazo substituent. Light irradiation of this compound triggers a network of E/Z isomerization and proton transfer reactions that enable autonomous and reversible ring shuttling away from equilibrium.
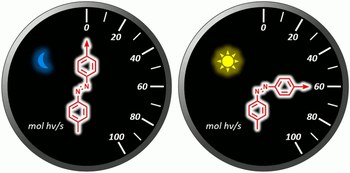
4,4′-Dimethylazobenzene as a chemical actinometer
Lorenzo Casimiro, Leonardo Andreoni, Jessica Groppi, Alberto Credi, Rémi Métivier, Serena Silvi
PHOTOCHEMICAL & PHOTOBIOLOGICAL SCIENCES
Published online: 16 January 2022
2022, Vol. 21, pp. 825-833
DOI: 10.1007/s43630-021-00162-3
Chemical actinometers are a useful tool in photochemistry, which allows to measure the photon flux of a light source to carry out quantitative analysis on photoreactions. The most commonly employed actinometers so far show minor drawbacks, such as difficult data treatment, parasite reactions, low stability or impossible reset. We propose herewith the use of 4,4′-dimethylazobenzene as a chemical actinometer. This compound undergoes a clean and efficient E/Z isomerization, approaching total conversion upon irradiation at 365 nm. Thanks to its properties, it can be used to determine the photon flux in the UV–visible region, with simple experimental methods and data treatment, and with the possibility to be reused after photochemical or thermal reset.
2021
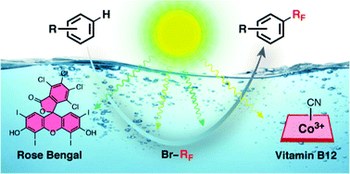
Bioinspired photocatalysed C-H fluoroalkylation of arenes in water promoted by native vitamin B12 and Rose Bengal
Damian E. Yerien, Al Postigo, Massimo Baroncini, Sebastian Barata-Vallejo
GREEN CHEMISTRY
Published online: 18 October 2021
2021, Vol. 23, pp. 8147-8153
DOI: 10.1039/d1gc02654b
The first perfluoroalkylation reaction of activated arenes (amino-substituted arenes and alkoxyarenes) has been achieved in water by employing the dyad Rose Bengal and hydrosoluble unmodified vitamin B-12 (cyanocobalamin) as the photocatalyst and co-catalyst, respectively. The reaction was performed under green LED irradiation and employed perfluoroalkyl bromides as the perfluoroalkyl radical sources. In particular, the presence of vitamin B-12 co-catalyst was found to be fundamental to elicit the formation of perfluoroalkyl radicals.
Photochemical energy conversion with artificial molecular machines
Leonardo Andreoni, Massimo Baroncini, Serena Silvi, Chiara Taticchi, Alberto Credi
ACS ENERGY & FUELS
Published online: 1 October 2021
2021, Vol. 35, pp. 18900-18914
DOI: 10.1021/acs.energyfuels.1c02921
The exploitation of sunlight as a clean, renewable, and distributed energy source is key to facing the energetic demand of modern society in a sustainable and affordable fashion. In the past few decades, chemists have learned to make molecular machines, that is, synthetic chemical systems in which energy inputs cause controlled movements of molecular components that could be used to perform a task. A variety of artificial molecular machines operated by light have been constructed by implementing photochemical processes within appropriately designed (supra)molecular assemblies. These studies could open up new routes for the realization of nanostructured devices and materials capable to harness, convert, and store light energy.
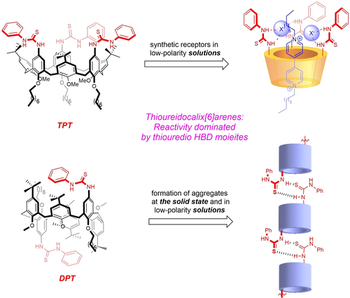
Thioureidocalix[6]arenes pseudorotaxanes
Gianpiero Cera, Margherita Bazzoni, Leonardo Andreoni, Federica Cester Bonati, Chiara Massera, Serena Silvi, Alberto Credi, Andrea Secchi, Arturo Arduini
EUROPEAN JOURNAL OF ORGANIC CHEMISTRY
Published online: 24 September 2021
2021, pp. 5788-5798
DOI: 10.1002/ejoc.202101080
Heteroditopic calix[6]arenes are extensively exploited as synthetic receptors and as molecular components of supramolecular threaded and interlocked structures. We describe the synthesis of two novel calix[6]arene macrocycles, the upper rim of which is functionalized with either two or three phenylthioureido groups. These moieties are excellent hydrogen-bond donors and largely affect the conformation of the macrocycle, both in solution and in the solid state. Alternate conformers are observed in solution, and they give rise to supramolecular chains in the solid state. Moreover, and because of this conformational freedom, also the recognition properties toward the formation of pseudorotaxanes with bipyridinium-based axles are strongly affected. The number of phenylthioureido units of the macrocyclic host and the nature of the counterions of the bipyridinium guest determine the stability of the complexes. NMR, UV-Vis and electrochemical measurements revealed that very stable 1 : 1 inclusion complexes are formed between the triphenylthioureido calix[6]arene and a bipyridinium guest with tosylate counterions, whereas no complex is observed between the diphenylthioureido macrocycle and a hexafluorophosphate bipyridinium salt.
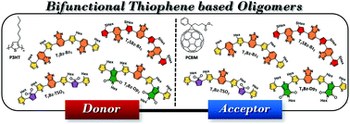
Push-pull thiophene-based small molecules with donor and acceptor units of varying strength for photovoltaic application: beyond P3HT and PCBM
Martina Marinelli, Andrea Candini, Filippo Monti, Alex Boschi, Mattia Zangoli, Elisabetta Salatelli, Filippo Pierini, Massimiliano Lanzi, Alberto Zanelli, Massimo Gazzano, Francesca Di Maria
JOURNAL OF MATERIALS CHEMISTRY C
Published online: 14 September 2021
2021, Vol. 9, pp. 11216-11228
DOI: 10.1039/d1tc02641k
Here is reported an expedient synthesis implementing enabling technologies of a family of thiophene-based heptamers alternating electron donor (D) and acceptor (A) units in a D-A '-D-A-D-A '-D sequence. The nature of the peripheral A groups (benzothiadiazole vs. thienopyrrole-dione vs. thiophene-S,S-dioxide) and the strength of the donor units (alkyl vs. thioalkyl substituted thiophene ring) have been varied to finely tune the chemical-physical properties of the D-A oligomers, to affect the packing arrangement in the solid-state as well as to enhance the photovoltaic performances. The optoelectronic properties of all compounds have been studied by means of optical spectroscopy, electrochemistry, and density functional theory calculations. Electrochemical measurements and Kelvin probe force microscopy (KPFM) predicted a bifunctional behaviour for these oligomers, suggesting the possibility of using them as donor materials when blended with PCBM, and as acceptor materials when coupled with P3HT. Investigation of their photovoltaic properties confirmed this unusual characteristic, and it is shown that the performance can be tuned by the different substitution pattern. Furthermore, thanks to their ambivalent character, binary non-fullerene small-molecule organic solar cells with negligible values of HOMO and LUMO offsets were also fabricated, resulting in PCEs ranging between 2.54-3.96%.
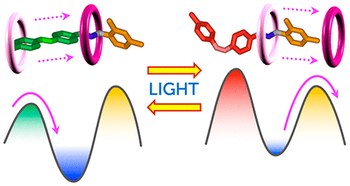
Second-generation light-fueled supramolecular pump
Martina Canton, Jessica Groppi, Lorenzo Casimiro, Stefano Corra, Massimo Baroncini, Serena Silvi, Alberto Credi
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY
Published online: 20 July 2021
2021, Vol. 143, pp. 10890-10894
DOI: 10.1021/jacs.1c06027
We describe the modular design of a pseudorotaxane-based supramolecular pump and its photochemically driven autonomous nonequilibrium operation in a dissipative regime. These properties derive from careful engineering of the energy maxima and minima along the threading coordinate and their light-triggered modulation. Unlike its precursor, this second-generation system is amenable to functionalization for integration into more complex devices.
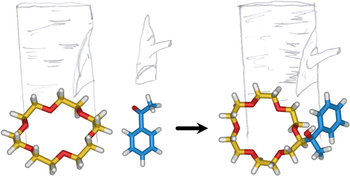
Direct synthetic routes to functionalised crown ethers
Federico Nicoli, Massimo Baroncini, Serena Silvi, Jessica Groppi, Alberto Credi
ORGANIC CHEMISTRY FRONTIERS
Published online: 1 July 2021
2021, Vol. 8, pp. 5531-5549
DOI: 10.1039/D1QO00699A
Crown ethers are macrocyclic hosts that can complex a wide range of inorganic and organic cations as well as neutral guest species. Their widespread utilization in several areas of fundamental and applied chemistry strongly relies on strategies for their functionalisation, in order to obtain compounds that could carry out multiple functions and could be incorporated in sophisticated systems. Although functionalised crown ethers are normally synthesised by templated macrocyclisation using appropriately substituted starting materials, the direct addition of functional groups onto a pre-formed macrocyclic framework is a valuable yet underexplored alternative. Here we review the methodologies for the direct functionalisation of aliphatic and aromatic crown ethers sporadically reported in the literature over a period of four decades. The general approach for the introduction of moieties on aliphatic crown ethers involves a radical mediated cross dehydrogenative coupling initiated either by photochemical or thermal/chemical activation, while aromatic crown ethers are commonly derivatised via electrophilic aromatic substitution. Direct functionalization routes can reduce synthetic effort, allow the later modification of crown ether-based architectures, and disclose new ways to exploit these versatile macrocycles in contemporary supramolecular science and technology.
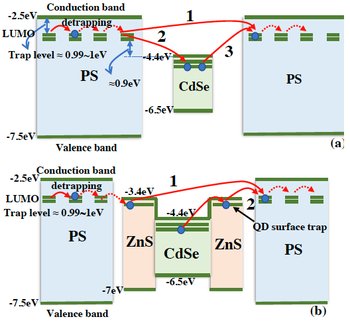
Space charge behaviour of quantum dot-doped polystyrene polymers
Zhipegn Lei, Davide Fabiani, Tommaso Bray, Chuanyang Li, Xinyu Wang, Thomas Andritsch, Alberto Credi, Marcello La Rosa
IEEE TRANSACTIONS ON DIELECTRICS AND ELECTRICAL INSULATION
Published online: June 2021
2021, Vol. 28, Issue 3, pp. 753-761
DOI: 10.1109/TDEI.2021.009223
This paper deals with the role played by the interface and bulk volume of the nanofiller about affecting the electrical properties of a nanocomposite material. For this purpose, a simple and completely amorphous matrix, polystyrene (PS), is used as base material, and core-shell quantum dots are exploited for simulating the structure of nanocomposites: CdSe core and CdSe-ZnS core-shell semiconductor quantum dots (QDs) are added into a PS matrix. The latter is to highlight the effect of the ZnS interface and as contrast to the core material. Dispersion and distribution of QDs are first microscopically observed and optimized, by including isopropyl alcohol in the manufacturing phase as an additional solvent. Among electrical properties the focus is on space charge accumulation, tested by means of the pulsed electroacoustic technique at 10 kV/mm and 50 kV/mm on CdSe and CdSe-ZnS doped PS composites. Results are then compared with a reference PS without QDs. Trap depth and density are also obtained by space charge measurement results. When CdSe QDs are added to PS, the trap density increases with respect to the baseline values measured on the unfilled polymer. In contrast, the ZnS shell around the CdSe core creates an additional trap level with lower trap depth, which increases charge mobility, thus turning homocharge into heterocharge accumulation. Therefore, the surface shell-structure of QD nanocrystals appears to significantly influence the space charge behavior of the nanocomposite, independently of the polymer.

Artificial supramolecular pumps powered by light
Stefano Corra, Lorenzo Casimiro, Massimo Baroncini, Jessica Groppi, Marcello La Rosa, Marina Tranfic Bakic, Serena Silvi, Alberto Credi
CHEMISTRY - A EUROPEAN JOURNAL
Published online: 5 May 2021
2021, Vol. 27, pp. 11076-11083
DOI: 10.1002/chem.202101163
* VIP *
The development of artificial nanoscale motors that can use energy from a source to perform tasks requires systems capable of performing directionally controlled molecular movements and operating away from chemical equilibrium. Here we describe the design, synthesis and properties of pseudorotaxanes in which a photon input triggers the unidirectional motion of a macrocyclic ring with respect to a non‐symmetric molecular axle. The photoinduced energy ratcheting at the basis of the pumping mechanism is validated by measuring the relevant thermodynamic and kinetic parameters. Owing to the photochemical behaviour of the azobenzene moiety embedded in the axle, the pump can repeat its operation cycle autonomously under continuous illumination. We use NMR spectroscopy to observe the dissipative non‐equilibrium state generated in situ by light irradiation. We also show that fine changes in the axle structure lead to an improvement in the performance of the motor. Such results highlight the modularity and versatility of this minimalist pump design, which provides facile access to dynamic systems that operate under photoinduced non‐equilibrium regimes.
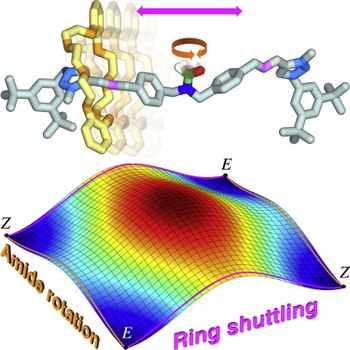
Stereodynamics of E/Z isomerization in rotaxanes through mechanical shuttling and covalent bond rotation
Stefano Corra, Christiaan de Vet, Massimo Baroncini, Alberto Credi, Serena Silvi
CHEM
Published online: 17 May 2021
2021, Vol. 7, pp. 2137-2150
DOI: 10.1016/j.chempr.2021.04.010
The mechanical bond has opened a new world for structural and dynamic stereochemistry, which is still largely underexplored and whose significance for various applications is becoming increasingly evident. We demonstrate that designed rearrangements involving both covalent and mechanical bonds can be integrated in [2]rotaxanes, leading to interesting consequences in terms of E/Z isomerization mechanisms. Two entirely distinct and concomitant stereomutations, pertaining to the same stereogenic element but involving different kinds of linkages within the molecule, are observed and are thoroughly characterized. The rate of the two processes is affected in opposite ways upon changing solvent polarity; such a phenomenon can be used to selectively modify the rate of each motion and adjust the relative contribution of the two mechanisms to the isomerization. Although the movements are not synchronized, an analysis of the intriguing fundamental implications for transition state theory, reaction pathway bifurcation, and microscopic reversibility was triggered by our experimental observations.
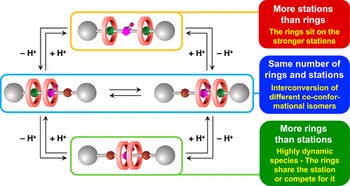
Chemically induced mismatch of rings and stations in [3]rotaxanes
Massimiliano Curcio, Federico Nicoli, Erica Paltrinieri, Ettore Fois, Gloria Tabacchi, Luigi Cavallo, Serena Silvi, Massimo Baroncini, Alberto Credi
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY
Published online: 29 April 2021
2021, Vol. 143, pp. 8046-8055
DOI: 10.1021/jacs.1c02230
The mechanical interlocking of molecular components can lead to the appearance of novel and unconventional properties and processes, with potential relevance for applications in nanoscience, sensing, catalysis, and materials science. We describe a [3]rotaxane in which the number of recognition sites available on the axle component can be changed by acid–base inputs, encompassing cases in which this number is larger, equal to, or smaller than the number of interlocked macrocycles. These species exhibit very different properties and give rise to a unique network of acid–base reactions that leads to a fine pKa tuning of chemically equivalent acidic sites. The rotaxane where only one station is available for two rings exhibits a rich coconformational dynamics, unveiled by an integrated experimental and computational approach. In this compound, the two crown ethers compete for the sole recognition site, but can also come together to share it, driven by the need to minimize free energy without evident inter-ring interactions.
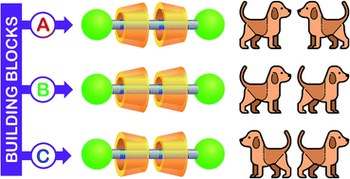
Selective access to constitutionally identical, orientationally isomeric calix[6]arene-based [3]rotaxanes by an active template approach
Margherita Bazzoni, Leonardo Andreoni, Serena Silvi, Alberto Credi, Gianpiero Cera, Andrea Secchi, Arturo Arduini
CHEMICAL SCIENCE
Published online: 1 April 2021
2021, Vol. 12, pp. 6419-6428
DOI: 10.1039/d1sc00279a
Tris(phenylureido)calix[6]arene is endowed with unique properties that make it a valuable macrocyclic component for the synthesis of mechanically interlocked molecules. Its three-dimensional and intrinsically nonsymmetric structure is kinetically selective toward two processes: (i) in apolar media, the threading of bipyridinium based axle-like components takes place exclusively from the upper rim; (ii) SN2 alkylation reactions of a pyridylpyridinium precursor engulfed in the cavity occur selectively at pyridylpyridinium nitrogen atom located at the macrocycle upper rim (active template synthesis). Here we exploit such properties to prepare two series of [3]rotaxanes, each consisting of three sequence isomers that arise from the threading of two identical but nonsymmetric wheels on a symmetric thread differing only for the reciprocal orientation of the macrocycles. The features of the calix[6]arene and the active template synthetic approach, together with a careful selection of the precursors, enabled us to selectively synthesise the [3]rotaxane sequence isomers of each series with fast kinetics and high yields.
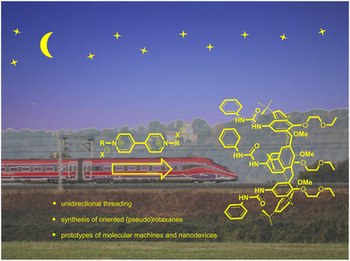
Heteroditopic calix[6]arene based intervowen and interlocked molecular devices
Gianpiero Cera, Arturo Arduini, Andrea Secchi, Alberto Credi, Serena Silvi
THE CHEMICAL RECORD
Published online: 3 March 2021
2021, Vol. 21, pp. 1161-1181
DOI: 10.1002/tcr.202100012
This personal account summarizes the achievement gained during the last two decades on the use of the calix[6]arene as a platform to build‐up interwoven and interlocked structures for the synthesis of oriented (pseudo)rotaxanes. We also account on how these calix[6]arene hosts affect the reactivity or spectroscopic properties of their bound guests.
Ruthenium tris(bipyridine) complexes: Interchange between photons and electrons in molecular-scale devices and machines
Vincenzo Balzani, Paola Ceroni, Alberto Credi, Margherita Venturi
COORDINATION CHEMISTRY REVIEWS
Published online: 15 January 2021
2021, Vol. 433, art. n. 213758
DOI: 10.1016/j.ccr.2020.213758
This review deals with a very peculiar molecule, [Ru(bpy)3]2+ (bpy: 2,2′-bipyridine), and its interaction with photons and electrons. We summarize the properties that make [Ru(bpy)3]2+ and related compounds a unique family of “clever” molecules which find application as components of chemical species capable of processing optical and electrical signals to perform a variety of interesting functions. Examples discussed in the article, that showcase the realization of these concepts with molecular and supramolecular systems, include molecular wires, switches, antennas and mechanical machines.

Out-of-equilibrium threaded and interlocked molecular structures
Massimo Baroncini, Serena Silvi, Alberto Credi
in OUT-OF-EQUILIBRIUM (SUPRA)MOLECULAR SYSTEMS AND MATERIALS (Eds: N. Giuseppone, A. Walther), Wiley-VCH, Weinheim
Published online: April 2021
Chapter 11, pp. 305-336
ISBN: 978-3-527-34615-8
The bottom-up design, construction, and operation of artificial mechanical devices and machines on the molecular scale is a topic of great interest in nanoscience and a fascinating challenge of nanotechnology. Biomolecular motors, executing crucial functions within living cells, demonstrate the utility and feasibility of systems of this kind. In recent years, the progress in several areas of chemistry, together with an improved understanding of biomolecular machines, has led to the development of a large variety of wholly synthetic molecular devices and machines that, in some cases, are able to perform specific tasks. These systems have the potential to bring about radical innovations in several areas of technology and medicine.
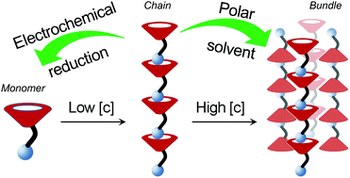
Hierarchical self-assembly and controlled disassembly of a cavitand-based host-guest supramolecular polymer
Daniele Zuccaccia, Roberta Pinalli, Rita De Zorzi, Monica Semeraro, Alberto Credi, Cristiano Zuccaccia, Alceo Macchioni, Silvano Geremia, Enrico Dalcanale
POLYMER CHEMISTRY
Published online: 26 November 2020
2021, Vol. 12, pp. 389-401
DOI: 10.1039/D0PY01483D
There is considerable interest in dynamic materials featuring modular components with nano-scale dimensions and controlled responsiveness to external stimuli. Supramolecular polymers are a class of materials that fulfil all these conditions well. Here, we present a family of host–guest supramolecular polymers that combine the outstanding complexing properties of tetraphosphonate cavitands toward N-methylpyridinium guests with molecular switching. The designed monomer is a cavitand featuring four inward facing P[double bond, length as m-dash]O groups at the upper rim and a single N-methylpyridinium unit at the lower rim, forming instantaneously a polymeric species in solution, thanks to the high complexation constants measured for these host–guest interactions. This system has been analyzed by NMR spectroscopy and electrochemical techniques. In order to interpret the results of diffusion-sensitive experiments, we took advantage of the X-ray crystal structure obtained for the polymeric species and developed an original treatment for the PGSE data by non-linear fitting. The analysis of the experimental data identified an isodesmic polymerization model at a monomer concentration below 20 mM, driven by intrachain host–guest interactions, and an additional level of tetrameric bundle aggregation above 20 mM, due to interchain dipolar and quadrupolar interactions. Two orthogonal disassembly procedures have been implemented: electrochemical reduction for the linear chains and solvent-driven dissolution for the bundles.
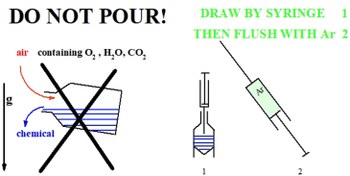
Manipulating chemicals at small- and semimicro-scale in inert atmosphere
Bruno Lunelli, Massimo Baroncini
CHEMISTRY-METHODS
Published online: 13 August 2021
2021, Vol. 1, pp. 421-426
DOI: 10.1002/cmtd.202100036
The rational use of chemicals requires that their properties be compatible with the planned application. These properties are modified by the interactions to which the chemical substance is subjected, among which particularly relevant are those with the components of the lab atmosphere, collectively labelled air sensitivity. The air sensitivity of chemicals is remarkably amplified in the presently common small- and semimicro-scale samples and operations, due to the increased surface-to-mass ratio with respect to the time-honoured multigram scale. In this note we present ergonomy-aware procedures implemented in small- and semimicro-scale laboratory operations, which enable to work easily in an inert atmosphere without the need for awkward or expensive apparatuses such as glove box or vacuum line.
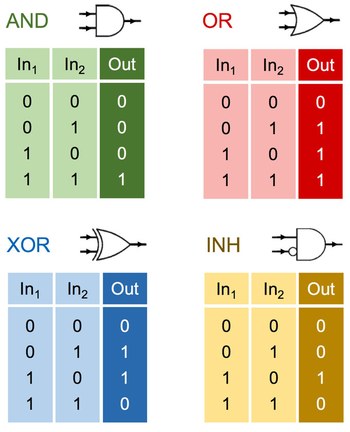
Binary logic operations with artificial molecular machines
Federico Nicoli, Erica Paltrinieri, Marina Tranfic Bakic, Massimo Baroncini, Serena Silvi, Alberto Credi
COORDINATION CHEMISTRY REVIEWS
Published online: 13 October 2020
2021, Vol. 428, art. n. 213589
DOI: 10.1016/j.ccr.2020.213589
Living systems rely on ions and molecules for gathering, elaborating and storing information, as well as for controlling motion across different length scales. Artificial mimics of such biomolecular systems, i.e., logic devices and mechanical machines, have been developed in the past decades. Here we discuss their combination to produce logic-controlled nanomachines, a research that holds great potential for basic science and prospective applications. Systems of this kind may be forerunners of ‘molecular robots’ that can execute specific functions as a result of gathering and processing multiple environmental stimuli.
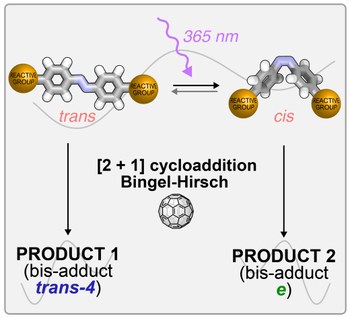
Light-controlled regioselective synthesis of fullerene bis-adducts
Luka Ðorđević, Lorenzo Casimiro, Nicola Demitri, Massimo Baroncini, Serena Silvi, Francesca Arcudi, Alberto Credi, Maurizio Prato
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 28 July 2020
2021, Vol. 60, pp. 313-320
DOI: 10.1002/anie.202009235
Multi-functionalization and isomer-purity of fullerenes are crucial tasks for the development of fullerene chemistry in various fields. In both current main approaches – tether-directed covalent functionalization and supramolecular masks – the control of regioselectivity requires multi-step synthetic procedures to prepare the desired tether or mask. Herein, we describe light-responsive tethers, containing an azobenzene photoswitch and two malonate groups, in the double cyclopropanation of [60]fullerene. The formation of the bis-adducts and their spectroscopic and photochemical properties, as well as the effect of azobenzene photoswitching on the regiochemistry of the bis-addition, have been studied. The behavior of the tethers depends on the geometry of the connection between the photoactive core and the malonate moieties. One tether lead to strikingly different adduct distribution for the E and Z isomers, indicating that the covalent bis-functionalization of C60 can be controlled by light.
2020
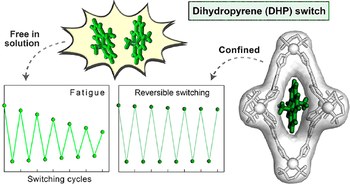
Improving fatigue resistance of dihydropyrene by encapsulation within a coordination cage
Martina Canton, Angela B. Grommet, Luca Pesce, Julius Gemen, Shiming Li, Yael Diskin-Posner, Alberto Credi, Giovanni M. Pavan, Joakim Andréasson, Rafal Klajn
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY
Published online: 14 August 2020
2020, Vol. 142, pp. 14557-14565
DOI: 10.1021/jacs.0c06146
Photochromic molecules undergo reversible isomerization upon irradiation with light at different wavelengths, a process that can alter their physical and chemical properties. For instance, dihydropyrene (DHP) is a deep-colored compound that isomerizes to light-brown cyclophanediene (CPD) upon irradiation with visible light. CPD can then isomerize back to DHP upon irradiation with UV light or thermally in the dark. Conversion between DHP and CPD is thought to proceed via a biradical intermediate; bimolecular events involving this unstable intermediate thus result in rapid decomposition and poor cycling performance. Here, we show that the reversible isomerization of DHP can be stabilized upon confinement within a PdII6L4 coordination cage. By protecting this reactive intermediate using the cage, each isomerization reaction proceeds to higher yield, which significantly decreases the fatigue experienced by the system upon repeated photocycling. Although molecular confinement is known to help stabilize reactive species, this effect is not typically employed to protect reactive intermediates and thus improve reaction yields. We envisage that performing reactions under confinement will not only improve the cyclic performance of photochromic molecules, but may also increase the amount of product obtainable from traditionally low-yielding organic reactions.
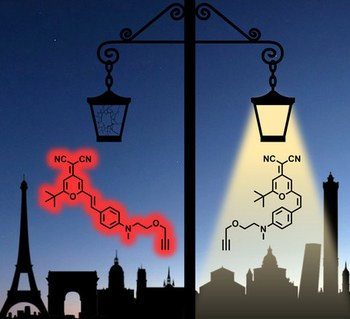
Photophysical properties of 4‐dicyanomethylene‐2‐methyl‐6‐(p‐dimethylamino‐styryl)‐4H‐pyran revisited: fluorescence versus photoisomerization
Lorenzo Casimiro, Stéphane Maisonneuve, Pascal Retailleau, Serena Silvi, Juan Xie, Rémi Métivier
CHEMISTRY - A EUROPEAN JOURNAL
Published online: 11 July 2020
2020, Vol. 26, pp. 14341-14350
DOI: 10.1002/chem.202002828
Although 4‐dicyanomethylene‐2‐methyl‐6‐(p‐dimethylamino‐styryl)‐4H‐pyran (DCM) has been known for many decades as a bright and photostable fluorophore, used for a wide variety of applications in chemistry, biology and physics, only little attention has been paid so far to the presence of multiple isomers and conformers, namely s‐trans‐(E), s‐cis‐(E), s‐trans‐(Z), and s‐cis‐(Z). In particular, light‐induced E–Z isomerization plays a great role on the overall photophysical properties of DCM. Herein, we give a full description of a photoswitchable DCM derivative by a combination of structural, theoretical and spectroscopic methods. The main s‐trans‐(E) isomer is responsible for most of the fluorescence features, whereas the s‐cis‐(E) conformer only contributes marginally. The non‐emitting Z isomers are generated in large conversion yields upon illumination with visible light (e.g., 485 or 514 nm) and converted back to the E forms by UV irradiation (e.g., 365 nm). Such photoswitching is efficient and reversible, with high fatigue resistance. The E→Z and Z→E photoisomerization quantum yields were determined in different solvents and at different irradiation wavelengths. Interestingly, the fluorescence and photoisomerization properties are strongly influenced by the solvent polarity: the fluorescence is predominant at higher polarity, whereas photoisomerization becomes more efficient at lower polarity. Intermediate medium (THF) represents an optimized situation with a good balance between these two features.
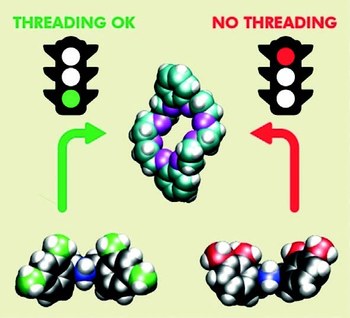
Precision molecular threading/dethreading
Jessica Groppi, Lorenzo Casimiro, Martina Canton, Stefano Corra, Mina Jafari-Nasab, Gloria Tabacchi, Luigi Cavallo, Massimo Baroncini, Serena Silvi, Ettore Fois, Alberto Credi
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 12 May 2020
2020, Vol. 59, pp. 14825-14834
DOI: 10.1002/anie.202003064
The general principles guiding the design of molecular machines based on interlocked structures are well known. Nonetheless, the identification of suitable molecular components for a precise tuning of the energetic parameters that determine the mechanical link is still challenging. Indeed, what are the reasons of the “all‐or‐nothing” effect, which turns a molecular “speed‐bump” into a stopper in pseudorotaxane‐based architectures? Here we investigate the threading and dethreading processes for a representative class of molecular components, based on symmetric dibenzylammonium axles and dibenzo[24]crown‐8 ether, with a joint experimental‐computational strategy. From the analysis of quantitative data and an atomistic insight, we derive simple rules correlating the kinetic behaviour with the substitution pattern, and provide rational guidelines for the design of modules to be integrated in molecular switches and motors with sophisticated dynamic features.

Manufacturing at nanoscale: from molecular machines to transducers
Marcello La Rosa, Massimo Baroncini, Serena Silvi, Alberto Credi
in ADVANCES IN NANOSTRUCTURED MATERIALS AND NANOPATTERNING TECHNOLOGIES (Eds: M. L. Focarete, V. Guarino, D. Pisignano) Elsevier, Amsterdam
Published online: 2020
Chapter 2, pp. 41-63
DOI: 10.1016/B978-0-12-816865-3.00002-0
The bottom-up design, construction, and operation of artificial mechanical devices and machines on the molecular scale is a topic of great interest in nanoscience and a fascinating challenge of nanotechnology. Biomolecular motors, executing crucial functions within living cells, demonstrate the utility and feasibility of systems of this kind. In recent years, the progress in several areas of chemistry, together with an improved understanding of biomolecular machines, has led to the development of a large variety of wholly synthetic molecular devices and machines that, in some cases, are able to perform specific tasks. These systems have the potential to bring about radical innovations in several areas of technology and medicine.
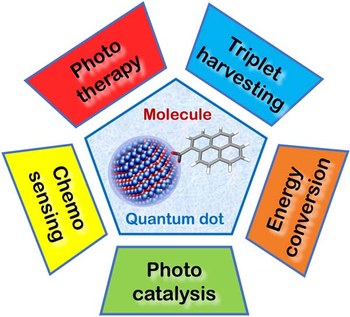
Semiconductor quantum dots as components of photoactive supramolecular architectures
Marcello La Rosa, Emily H. Payne, Alberto Credi
CHEMISTRYOPEN
Published online: 10 February 2020
2020, Vol. 9, Issue 2, pp. 200-213
DOI: 10.1002/open.201900336
Luminescent quantum dots (QDs) are colloidal semiconductor nanocrystals consisting of an inorganic core covered by a molecular layer of organic surfactants. Although QDs have been known for more than thirty years, they are still attracting the interest of researchers because of their unique size‐tunable optical and electrical properties arising from quantum confinement. Moreover, the controlled decoration of the QD surface with suitable molecular species enables the rational design of inorganic‐organic multicomponent architectures that can show a vast array of functionalities. This minireview highlights the recent progress in the use of surface‐modified QDs – in particular, those based on cadmium chalcogenides – as supramolecular platforms for light‐related applications such as optical sensing, triplet photosensitization, photocatalysis and phototherapy.
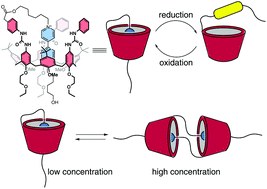
Synthesis and properties of a redox-switchable calix[6]arene-based molecular lasso
Guido Orlandini, Lorenzo Casimiro, Margherita Bazzoni, Beatrice Cogliati, Alberto Credi, Marco Lucarini, Serena Silvi, Arturo Arduini, Andrea Secchi
ORGANIC CHEMISTRY FRONTIERS
Published online: 30 December 2019
2020, Vol. 7, pp. 648-659
DOI: 10.1039/c9qo01379b
The synthesis and characterisation of calix[6]arene-based lasso-like molecular structures is described. These interwoven structures consist of an electrochemical responsive N,N′-dialkylviologen arm covalently anchored at the upper rim of a triphenylureido calix[6]arene-based wheel. Upon reduction of the viologen core, a hollow tridimensional macrocyclic structure can be generated. This process is reversible, and the original lasso-like structure can be regenerated by oxidizing the viologen arm to its original dicationic form. Electrochemical and EPR techniques investigated the ability of the system to perform threading/dethreading movements upon redox switching. The functionalisation of the arm ω-hydroxy ending with a bulky diphenylacetyl group converts the self-threaded structure in a blocked interwoven molecular compound belonging to the class of [1]rotaxanes. The ability to form dimeric structures in the shape of a [c2]daisy chain was also demonstrated, an unprecedented result for calixarene macrocycles.

Macchine molecolari
Alberto Credi, Vincenzo Balzani
ITHACA - VIAGGIO NELLA SCIENZA
Published online: 1 January 2020
2020, Vol. XIV, pp. 5-17
La costruzione di macchine di dimensioni molecolari è uno straordinario risultato scientifico e un obiettivo primario della nanotecnologia. Nel corso degli ultimi trent’anni, i chimici in varie parti del mondo hanno imparato a realizzare delle semplici macchine nanometriche. Anche se questi minuscoli dispositivi non sono ancora divenuti parte integrante della nostra vita quotidiana, siamo ormai alle soglie di una nuova rivoluzione industriale, capace di cambiare il nostro futuro con applicazioni innovative nella tecnologia dei materiali, nell’informatica, nella robotica e nella medicina.
Photo-activated artificial molecular machines that can perform tasks
Stefano Corra, Massimiliano Curcio, Massimo Baroncini, Serena Silvi, Alberto Credi
ADVANCED MATERIALS
Published online: 20 January 2020
2020, Vol. 32, Issue 20, art. n. 1906064, pp. 1-22
DOI: 10.1002/adma.201906064
Research on artificial photoactivated molecular machines has moved in recent years from a basic scientific endeavor toward a more applicative effort. Nowadays, the prospect of reproducing the operation of natural nanomachines with artificial counterparts is no longer a dream but a concrete possibility. The progress toward the construction of molecular‐machine‐based devices and materials in which light irradiation results in the execution of a task as a result of nanoscale movements is illustrated here. After a brief description of a few basic types of photoactivated molecular machines, significant examples of their exploitation to perform predetermined functions are presented. These include switchable catalysts, nanoactuators that interact with cellular membranes, transporters of small molecular cargos, and active joints capable of mechanically coupling molecular‐scale movements. Investigations aimed at harnessing the collective operation of a multitude of molecular machines organized in arrays to perform tasks at the microscale and macroscale in hard and soft materials are also reviewed. Surfaces, gels, liquid crystals, polymers, and self‐assembled nanostructures are described wherein the nanoscale movement of embedded molecular machines is amplified, allowing the realization of muscle‐like actuators, microfluidic devices, and polymeric materials for light energy transduction and storage.
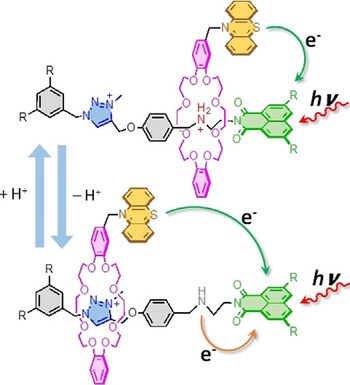
Photoinduced electron transfer involving a naphthalimide chromophore in switchable and flexible [2]rotaxanes
Benoit Colasson, Alberto Credi, Barbara Ventura
CHEMISTRY - A EUROPEAN JOURNAL
Published online: 22 October 2019
2020, Vol. 26, Issue 2, pp. 534-542
DOI: 10.1002/chem.201904155
The interlocking of ring and axle molecular components in rotaxanes provides a way to combine chromophoric, electron‐donor and electron‐acceptor moieties in the same molecular entity, in order to reproduce the features of photosynthetic reaction centers. To this aim, the photoinduced electron transfer processes involving a 1,8‐naphthalimide chromophore, embedded in several rotaxane‐based dyads, were investigated by steady‐state and time‐resolved absorption and luminescence spectroscopic experiments in the 300 fs–10 ns time window. Different rotaxanes built around the dialkylammonium/ dibenzo[24]crown‐8 ether supramolecular motif were designed and synthesized to decipher the relevance of key structural factors, such as the chemical deactivation of the ammonium‐crown ether recognition, the presence of a secondary site for the ring along the axle, and the covalent functionalization of the macrocycle with a phenothiazine electron donor. Indeed, the conformational freedom of these compounds gives rise to a rich dynamic behavior induced by light and may provide opportunities for investigating and understanding phenomena that take place in complex (bio)molecular architectures.
Photo- and redox-driven artificial molecular motors
Massimo Baroncini, Serena Silvi, Alberto Credi
CHEMICAL REVIEWS
Published online: 15 August 2019
2020, Vol.120, pp. 200-268
DOI: 10.1021/acs.chemrev.9b00291
Directed motion at the nanoscale is a central attribute of life, and chemically driven motor proteins are nature’s choice to accomplish it. Motivated and inspired by such bionanodevices, in the past few decades chemists have developed artificial prototypes of molecular motors, namely, multicomponent synthetic species that exhibit directionally controlled, stimuli-induced movements of their parts. In this context, photonic and redox stimuli represent highly appealing modes of activation, particularly from a technological viewpoint. Here we describe the evolution of the field of photo- and redox-driven artificial molecular motors, and we provide a comprehensive review of the work published in the past 5 years. After an analysis of the general principles that govern controlled and directed movement at the molecular scale, we describe the fundamental photochemical and redox processes that can enable its realization. The main classes of light- and redox-driven molecular motors are illustrated, with a particular focus on recent designs, and a thorough description of the functions performed by these kinds of devices according to literature reports is presented. Limitations, challenges, and future perspectives of the field are critically discussed.
2019
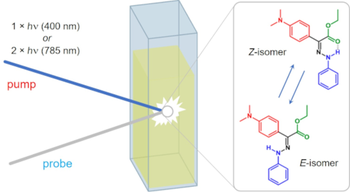
Ultrafast processes triggered by one- and two-photon excitation of a photochromic and luminescent hydrazone
Alessandro Iagatti, Baihao Shao, Alberto Credi, Barbara Ventura, Ivan Aprahamian, Mariangela Di Donato
BEILSTEIN JOURNAL OF ORGANIC CHEMISTRY
Published online: 15 October 2019
2019, Vol. 15, pp. 2438-2446
DOI: 10.3762/bjoc.15.236
In this work we apply a combination of steady state and time resolved luminescence and absorption spectroscopies to investigate the excited-state dynamics of a recently developed molecular photoswitch, belonging to the hydrazone family. The outstanding properties of this molecule, involving fluorescence toggling, bistability, high isomerization quantum yield and non-negligible two-photon absorption cross section, make it very promising for numerous applications. Here we show that the light induced Z/E isomerization occurs on a fast <1 ps timescale in both toluene and acetonitrile, while the excited state lifetime of the Z-form depends on solvent polarity, suggesting a partial charge transfer nature of its low lying excited state. Time-resolved luminescence measurements evidence the presence of a main emission component in the 500–520 nm spectral range, attributed to the Z-isomer, and a very short living blue-shifted emission, attributed to the E-isomer. Finally, transient absorption measurements performed upon far-red excitation are employed as an alternative method to determine the two-photon absorption cross-section of the molecule.

Design of photo-activated molecular machines: highlights from the past ten years
Jessica Groppi, Massimo Baroncini, Margherita Venturi, Serena Silvi, Alberto Credi
CHEMICAL COMMUNICATIONS
Published online: 25 September 2019
2019, Vol. 55, pp. 12595-12602
DOI: 10.1039/c9cc06516d
Molecular machines powered by light are appropriately designed multicomponent molecular structures that exploit photochemical processes to perform controlled movements. We discuss the advances in the design and investigation of light-driven molecular machines by presenting a selection of examples from the past decade.
Individual‐molecule perspective analysis of chemical reaction networks: the case of a light‐driven supramolecular pump
Andrea Sabatino, Emanuele Penocchio, Giulio Ragazzon, Alberto Credi, Diego Frezzato
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 4 August 2019
2019, Vol. 58, pp. 14341-14348
DOI: 10.1002/anie.201908026
The first study in which stochastic simulations of a two‐component molecular machine are performed in the mass‐action regime is presented. This system is an autonomous molecular pump consisting of a photoactive axle that creates a directed flow of rings through it by exploiting light energy away from equilibrium. The investigation demonstrates that the pump can operate in two regimes, both experimentally accessible, in which light‐driven steps can be rate‐determining or not. The number of photons exploited by an individual molecular pump, as well as the precision of cycling and the overall efficiency, critically rely on the operating regime of the machine. This approach provides useful information not only to guide the chemical design of a self‐assembling molecular device with desired features, but also to elucidate the effect of the environment on its performance, thus facilitating its experimental investigation.

Light on molecular devices
Alberto Credi
CHEMPHOTOCHEM
Published online: 24 June 2019
2019, Vol. 3, pp. 578-579
DOI: 10.1002/cptc.201900172
Meet the board: Alberto Credi is a Professor of Chemistry at the Università di Bologna (Italy) and an Associate Research Director at the Consiglio Nazionale delle Ricerche. He is a current member of the ChemPhotoChem Editorial Board.
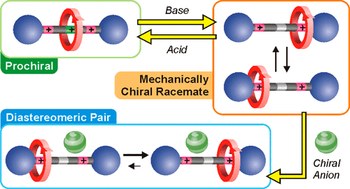
Chemical on/off switching of mechanically planar chirality and chiral anion recognition in a [2]rotaxane molecular shuttle
Stefano Corra, Christiaan de Vet, Jessica Groppi, Marcello La Rosa, Serena Silvi, Massimo Baroncini, Alberto Credi
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY
Published online: 25 May 2019
2019, Vol. 141, pp. 9129-9133
DOI: 10.1021/jacs.9b00941
We exploit a reversible acid-base triggered molecular shuttling process to switch an appropriately designed rotaxane between prochiral and mechanically planar chiral forms. The mechanically planar enantiomers and their interconversion, arising from ring shuttling, have been characterized by NMR spectroscopy. We also show that the supramolecular interaction of the positively charged rotaxane with optically active anions causes an imbalance in the population of the two enantiomeric coconformations. This result represents an unprecedented example of chiral molecular recognition and can disclose innovative approaches to enantioselective sensing and catalysis.

Light-responsive (supra)molecular architectures: recent advances
Massimo Baroncini, Jessica Groppi, Stefano Corra, Serena Silvi, Alberto Credi
ADVANCED OPTICAL MATERIALS
First published: 17 May 2019
2019, Vol. 7, Issue 16, art. n.1900392, pp. 1-16
DOI: 10.1002/adom.201900392
The development and investigation of (supra)molecular‐based architectures characterized by light‐activated functionalities is a highly relevant topic of chemical research. The interest on photo‐controlled systems arises not only from their potential applications in different fields of technology but also from their scientific significance related to the understanding of light–matter interactions at the nanoscale. Indeed, light is a peculiar and unique tool as it can be conveniently applied to supply the energy required to affect and operate a system and, at the same time, to probe its state and investigate its transformations. Some basic aspects of light‐induced processes in (supra)molecular architectures are discussed here in the frame of their use to implement novel functionalities in nanostructured systems and materials. In this perspective, a few recent examples from our own work will be illustrated which are meant to provide an overview of the current directions in this highly cross‐disciplinary field of research.
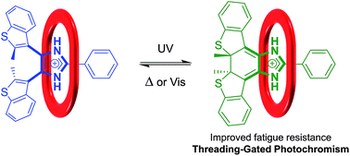
Threading-gated photochromism in [2]pseudorotaxanes
Giorgio Baggi, Lorenzo Casimiro, Massimo Baroncini, Serena Silvi, Alberto Credi, Stephen J. Loeb
CHEMICAL SCIENCE
Published online: 23 April 2019
2019, Vol. 10, pp. 5104-5113
DOI: 10.1039/c9sc00913b
Rigid, Y-shaped imidazole compounds containing the bis(thienyl)ethene moiety were designed and synthesized. The 4,5-bis(benzothienyl)-2-phenylimidazolium cations were then used as axles for [2]pseudorotaxane formation with 24-membered crown ether wheels. It was demonstrated using 1H NMR spectroscopy, UV-Vis absorption and emission spectroscopies that this host-guest interaction results in significant changes in the photochromic properties of the imidazolium axles. This is a rare example of gated photochromism, which exploits the recognition event of an interpenetrated molecular system to tune the photochromic properties in one of the components.
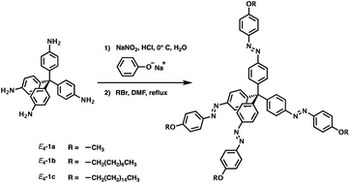
Solution and solid state photochromism in a family of shape persistent azobenzene tetramers functionalized with alkyloxy substituents
Jacopo Vialetto, Jessica Groppi, Marcello La Rosa, Serena Silvi, Alberto Credi, Massimo Baroncini
PHOTOCHEMICAL & PHOTOBIOLOGICAL SCIENCES
Published online: 10 April 2019
2019, Vol. 18, Issue 9, pp. 2281-2286
DOI: 10.1039/c9pp00022d
Shape-persistent azobenzene tetramers functionalized at the periphery with alkyloxy substituents of different length have been synthesized, and their photochemical behaviour has been investigated. Efficient E → Z photoisomerization of the azobenzene units takes place both in solution and in the solid state, a highly desirable yet uncommon property for azobenzene-type photochromic compounds. The solid state E → Z photoisomerization is accompanied by an isothermal crystal–amorphous phase transformation; successively, anisotropic crystals can be grown upon promoting the Z → E isomerization by thermal annealing of the irradiated samples. These results validate the strategy of engineering multiphotochromic architectures with a rigid star-shaped geometry to preserve the solution-based photoreactivity also in the solid state. The observed unexpected photoinduced alignment makes these materials potentially attractive for the development of photo-patternable and photo-responsive surfaces.
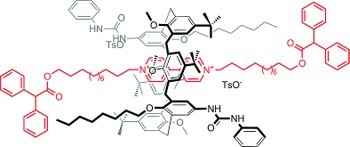
New geometries for calix[6]arene-based rotaxanes
Margherita Bazzoni, Valeria Zanichelli, Lorenzo Casimiro, Chiara Massera, Alberto Credi, Andrea Secchi, Serena Silvi, Arturo Arduini
EUROPEAN JOURNAL OF ORGANIC CHEMISTRY
Published online: 22 March 2019
2019, pp. 3513-3124
DOI: 10.1002/ejoc.201900211
Understanding the role played by the nature, number and arrangement of binding sites anchored to a macrocycle remains a topic of current interest for the synthesis of new interwoven species. We report the synthesis and detailed structural characterisation of a new calix[6]arene derivative decorated with two phenylureido groups anchored at the diametrical position of the calix upper rim that adopts a 1,2,3-alternate conformation in solution and in the solid state. Preliminary data on the ability of this host to form redox-active pseudorotaxanes and rotaxanes are reported.

A molecular cable car for transmembrane ion transport
Alberto Credi
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 20 February 2019
2019, Vol. 58, pp. 4108-4110
DOI: 10.1002/anie.201814333
The controlled transport of molecular and ionic substrates across bilayer membranes is a fundamental task for the operation of living organisms. It is also a highly fascinating and demanding challenge for artificial molecular machines. The recent report of a synthetic transmembrane molecular shuttle that can transport potassium ions selectively down a gradient in a liposomal system makes a small but significant step forward towards this goal.
EPR sensing of metal and organic cations using a novel spin-labelled dibenzo-24-crown-8-ether
Lorenzo Gualandi, Paola Franchi, Alberto Credi, Elisabetta Mezzina, Marco Lucarini
PHYSICAL CHEMISTRY CHEMICAL PHYSICS
Published online: 7 September 2018
2019, Vol. 21, pp. 3558-3563
DOI: 10.1039/C8CP04362K
The synthesis of novel dibenzo-24-crown ether substituted nitroxides and their use as spin probes for the detection of cation guests by EPR are reported. Formation of a host–guest complex between the proposed spin probes and several cations, both organic and inorganic, was evidenced by a significant change in the value of the benzylic and nitrogen EPR hyperfine splittings upon complexation. This favorable feature provided a reliable EPR sensor that is able to selectively distinguish different cationic guests.
2018
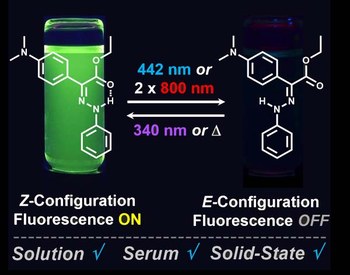
Solution and solid-state emission toggling of a photochromic hydrazone
Baihao Shao, Massimo Baroncini, Hai Qian, Laura Bussotti, Mariangela Di Donato, Alberto Credi, Ivan Aprahamian
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY
Published online: 25 September 2018
2018, Vol. 140, pp. 12323-12327
DOI: 10.1021/jacs.8b07108
The proliferation of light-activated switches in recent years has enabled their use in a broad range of applications encompassing an array of research fields and disciplines. All current systems, however, have limitations (e.g., from complicated synthesis to incompatibility in biologically relevant media and lack of switching in the solid-state) that can stifle their real-life application. Here we report on a system that packs most, if not all, the desired, targeted and sought-after traits from photochromic compounds (bistability, switching in various media ranging from serum to solid-state, while exhibiting ON/OFF fluorescence emission switching, and two-photon assisted near infrared light toggling) in an easily accessible structure.
Photoactive molecular-based devices, machines and materials: recent advances
Massimo Baroncini, Martina Canton, Lorenzo Casimiro, Stefano Corra, Jessica Groppi, Marcello La Rosa, Serena Silvi, Alberto Credi
EUROPEAN JOURNAL OF INORGANIC CHEMISTRY
Published online: 14 September 2018
2018, pp. 4589-4603
DOI: 10.1002/ejic.201800923
* VIP *
Molecular and supramolecular-based systems and materials that can perform predetermined functions in response to light stimulation have been extensively studied in the past three decades. Their investigation continues to be a highly stimulating topic of chemical research, not only because of the inherent scientific value related to a bottom-up approach to functional nanostructures, but also for the prospective applications in diverse fields of technology and medicine. Light is an important tool in this context, as it can be conveniently used both for supplying energy to the system and for probing its states and transformations. In this microreview we recall some basic aspects of light-induced processes in (supra)molecular assemblies, and discuss their exploitation to implement novel functionalities with nanostructured devices, machines and materials. To this aim we illustrate a few examples from our own recent work, which are meant to illustrate the trends of current research in the field.
Among the top 10% most downloaded articles of the journal in the 12 months following publication

Reversible photoswitching and isomer-dependent diffusion of single azobenzene tetramers on a metal surface
Christophe Nacci, Massimo Baroncini, Alberto Credi, Leonhard Grill
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 6 September 2018
2018, Vol. 57, pp 15034-15039
DOI: 10.1002/anie.201806536
Azobenzene is a prototypical molecular switch that can be reversibly photoisomerized between the nearly planar and apolar trans form, and the distorted, polar cis form. Thus far, most studies related to azobenzene derivatives have focused on planar adsorbed molecules. We present here the study of a three‐dimensional shape‐persistent molecular architecture consisting of four tetrahedrally arranged azobenzene units and adsorbed on a Ag(111) surface. While the azobenzenes of the tripod in contact with the surface lose their switching ability, different isomers of the upright standing arm of the tetramer are obtained reversibly and efficiently by illumination at different wavelengths, revealing time constants of only a few minutes. Diffusion on the surface turns out to be dependent on the isomeric state ‐ trans or cis ‐ of the upright oriented azobenzene group. Hence, molecular mobility can be modulated via their isomeric state, which suggests that for instance molecular growth processes could be controlled by external stimuli.
Electrochemically triggered co-conformational switching in a [2]catenane comprising a non-symmetric calix[6]arene wheel and a two-station oriented macrocycle
Valeria Zanichelli, Luca Dallacasagrande, Arturo Arduini, Andrea Secchi, Giulio Ragazzon, Serena Silvi, Alberto Credi
MOLECULES
Published online: 11 May 2018
2018, Vol. 23, Issue 5, art. n. 1156, pp. 1-12
DOI: 10.3390/molecules23051156
Catenanes with desymmetrized ring components can undergo co-conformational rearrangements upon external stimulation and can form the basis for the development of molecular rotary motors. We describe the design, synthesis and properties of a [2]catenane consisting of a macrocycle—the ‘track’ ring—endowed with two distinct recognition sites (a bipyridinium and an ammonium) for a calix[6]arene—the ‘shuttle’ ring. By exploiting the ability of the calixarene to thread appropriate non-symmetric axles with directional selectivity, we assembled an oriented pseudorotaxane and converted it into the corresponding oriented catenane by intramolecular ring closing metathesis. Cyclic voltammetric experiments indicate that the calixarene wheel initially surrounds the bipyridinium site, moves away from it when it is reduced, and returns in the original position upon reoxidation. A comparison with appropriate model compounds shows that the presence of the ammonium station is necessary for the calixarene to leave the reduced bipyridinium site.
Synthesis of densely-packaged, ultrasmall Pt(0)2 clusters within a thioether functionalised MOF and their catalytic activity in industrial reactions at low–temperature
Marta Mon, Miguel A. Rivero-Crespo, Jesús Ferrando-Soria, Alejandro Vidal-Moya, Mercedes Boronat, Antonio Leyva-Pérez, Avelino Corma, Juan C. Hernández-Garrido, Miguel López-Haro, José J. Calvino, Giulio Ragazzon, Alberto Credi, Donatella Armentano, Emilio Pardo
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 30 March 2018
2018, Vol. 57, pp. 6186-6191
DOI: 10.1002/anie.201801957
The gram-scale synthesis, stabilization, and characterization of well-defined ultrasmall subnanometric catalytic clusters on solids is a challenge. The chemical synthesis and X-ray snapshots of Pt02 clusters, homogenously distributed and densely packaged within the channels of a metal–organic framework, is presented. This hybrid material catalyzes efficiently, and even more importantly from an economic and environmental viewpoint, at low temperature (25 to 140 °C), energetically costly industrial reactions in the gas phase such as HCN production, CO2 methanation, and alkene hydrogenations. These results open the way for the design of precisely defined catalytically active ultrasmall metal clusters in solids for technically easier, cheaper, and dramatically less-dangerous industrial reactions.
Photochemical investigation of cyanoazobenzene derivatives as components of artificial supramolecular pumps
Lorenzo Casimiro, Jessica Groppi, Massimo Baroncini, Marcello La Rosa, Alberto Credi, Serena Silvi
PHOTOCHEMICAL & PHOTOBIOLOGICAL SCIENCES
Published online: 3 May 2018
2018, Vol. 17, Issue 6, pp. 734-740
DOI: 10.1039/c8pp00062j
Among the plethora of photochromes reported so far, azobenzene has been proven to be the most suitable photoswitch for molecular systems and materials, due to its highly efficient and clean E–Z photoisomerization. Here we report two ammonium-based molecular axles bearing one or two p-cyanoazobenzene units at the extremities, able to form pseudorotaxanes with a crown ether macrocycle. The photochemistry of these compounds was studied in the isolated forms and in the pseudorotaxanes, showing that the functionalization speeds up the threading process without affecting the photochemical properties of the system. These results suggest that the investigated pseudorotaxanes can form the basis of new prototypes of artificial molecular-level pumps.
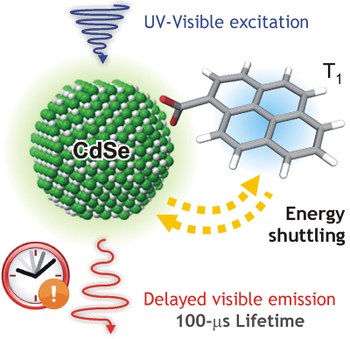
Designed long lived emission from CdSe quantum dots by reversible electronic energy transfer with a surface bound chromophore
Marcello La Rosa, Sergey A. Denisov, Gediminas Jonusauskas, Nathan D. McClenaghan, Alberto Credi
ANGEWANDTE CHEMIE INTERNATIONAL EDITION
Published online: 2 February 2018
2018, Vol. 57, pp. 3104-3107
DOI: 10.1002/anie.201712403
The size-tunable emission of luminescent quantum dots (QDs) makes them highly interesting for applications that range from bioimaging to optoelectronics. For the same applications, engineering their luminescence lifetime – in particular, making it longer – would be as important; however, no rational methodology to reach this goal is available to date. Here we describe a strategy to prolong the emission lifetime of QDs by electronic energy shuttling to the triplet excited state of a surface-bound molecular chromophore. To implement this idea we made CdSe QDs of different sizes, and we self-assembled them with a pyrene derivative. We observed that the conjugates exhibit delayed luminescence, with emission decays that are prolonged by more than 3 orders of magnitude compared to the parent CdSe QDs (lifetimes up to 330 us). The mechanism invokes unprecedented reversible quantum dot-to-organic chromophore electronic energy transfer.

Making and operating molecular machines: a multidisciplinary challenge
Massimo Baroncini, Lorenzo Casimiro, Christiaan de Vet, Jessica Groppi, Serena Silvi, Alberto Credi
CHEMISTRYOPEN
Published online: 2 February 2018
2018, Vol. 7, pp. 169-179
DOI: 10.1002/open.201700181
Movement is one of the central attributes of life, and a key feature in many technological processes. While artificial motion is typically provided by macroscopic engines powered by internal combustion or electrical energy, movement in living organisms is produced by machines and motors of molecular size that typically exploit the energy of chemical fuels at ambient temperature to generate forces and ultimately execute functions. The progress in several areas of chemistry, together with an improved understanding of biomolecular machines, has led to the development of a large variety of wholly synthetic molecular machines. These systems have the potential to bring about radical innovations in several areas of technology and medicine. In this article we discuss, with the help of a few examples, the multidisciplinary aspects of research on artificial molecular machines, and highlight its translational character.
Among the top 10% most downloaded articles of the journal in the 12 months following publication
2017
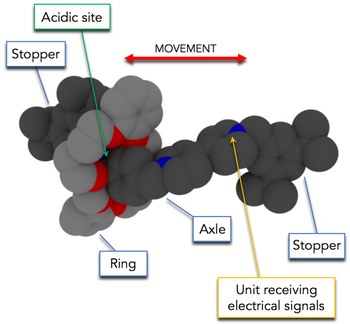
Remote electrochemical modulation of pKa in a rotaxane by co-conformational allostery
Giulio Ragazzon, Christian Schäfer, Paola Franchi, Serena Silvi, Benoit Colasson, Marco Lucarini, Alberto Credi
PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA
Published online: 27 November 2017
2018, Vol. 115, pp. 9385-9390
DOI: 10.1073/pnas.1712783115
Allosteric control, one of Nature’s most effective ways to regulate functions in biomolecular machinery, involves the transfer of information between distant sites. The mechanistic details of such a transfer are still an object of intensive investigation and debate, and the idea that intramolecular communication could be enabled by dynamic processes is gaining attention as a complement to traditional explanations. Mechanically interlocked molecules, owing to the particular kind of connection between their components and the resulting dynamic behavior, are attractive systems to investigate allosteric mechanisms and exploit them to develop functionalities with artificial species. We show that the pKa of an ammonium site located on the axle component of a [2]rotaxane can be reversibly modulated by changing the affinity of a remote recognition site for the interlocked crown ether ring through electrochemical stimulation. The use of a reversible ternary redox switch enables us to set the pKa to three different values, encompassing more than seven units. Our results demonstrate that in the axle the two sites do not communicate, and that in the rotaxane the transfer of information between them is made possible by the shuttling of the ring, that is, by a dynamic intramolecular process. The investigated coupling of electron- and proton-transfer reactions is reminiscent of the operation of the protein complex I of the respiratory chain.

An efficient method for the surface functionalization of luminescent quantum dots with lipoic acid-based ligands
Marcello La Rosa, Tommaso Avellini, Christophe Lincheneau, Serena Silvi, Iain A. Wright, Edwin C. Constable, Alberto Credi
EUROPEAN JOURNAL OF INORGANIC CHEMISTRY
Published online: 9 October 2017
2017, pp. 5143-5151
DOI: 10.1002/ejic.201700781
We describe herein an operationally advantageous general methodology for efficiently activating lipoic acid based compounds, a family of popular surface ligands for semiconductor nanocrystals, through the use of a borohydride exchange resin, and the use of the activated species to replace the native surface ligands of quantum dots. The procedure enabled phase transfer of the nanocrystals between polar and aqueous media and, if unsubstituted lipoic acid was used, a facile adjustment of their solubility in a wide range of solvents with varying polarity (from hexane to water). We show that the protocol is applicable to different types of nanocrystals and a variety of lipoic acid based ligands, and that the resulting quantum dots maintain their optical properties, in particular, an intense luminescence, and long-term colloidal stability.
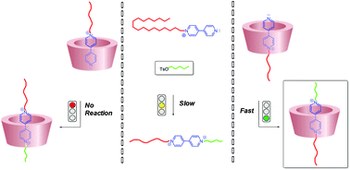
Efficient active-template synthesis of calix[6]arene-based oriented pseudorotaxanes and rotaxanes
Valeria Zanichelli, Giulio Ragazzon, Guido Orlandini, Margherita Venturi, Alberto Credi, Serena Silvi, Arturo Arduini, Andrea Secchi
ORGANIC & BIOMOLECULAR CHEMISTRY
Published online: 25 July 2017
2017, Vol. 15, pp. 6753-6763
DOI: 10.1039/C7OB01642E
A substrate can modify its chemical features, including a change of its reactivity, as a consequence of non-covalent interactions upon inclusion within a molecular host. Since the rise of supramolecular chemistry, this phenomenon has stimulated the ingenuity of scientists to emulate the function of enzymes by designing supramolecular systems in which the energetics and selectivity of reactions can be manipulated through programmed host–guest interactions and/or steric confinement. In this paper we investigate how the engulfment of a positively charged pyridinium-based guest inside the π-rich cavity of a tris-(N-phenylureido)calix[6]arene host affects its reactivity towards a SN2 reaction. We found that the alkylation of complexed substrates leads to the formation of pseudorotaxanes and rotaxanes with faster kinetics and higher yields with respect to the standard procedures exploited so far. More importantly, the strategy described here expands the range of efficient synthetic routes for the formation of mechanically interlocked species with a strict control of the mutual orientation of their non-symmetric molecular components.
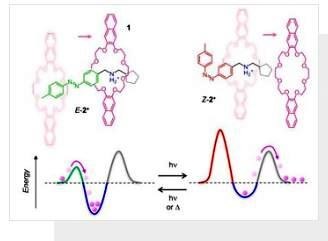
Processing light signals by molecular and supramolecular devices, machines and materials
Massimo Baroncini, Alberto Credi, Serena Silvi
EPA NEWSLETTER
June 2017, No. 92, pp. 30-34
Research in the past two decades has shown that chemical substrates can operate as input/output devices and are capable of handling chemical, electrical and optical signals. The conversion of the input(s) into the output(s) by the substrates, which include molecules, supramolecular systems, nanostructured and bulk materials, can give rise to functions spanning from chemosensing to smart drug delivery and mechanical actuation. Among the possible types of signals that can be processed by chemical substrates, light is certainly very attractive because it can be used both to write and read the system, it can be finely tuned in energy, time and space domains, and it is a potentially clean stimulus.
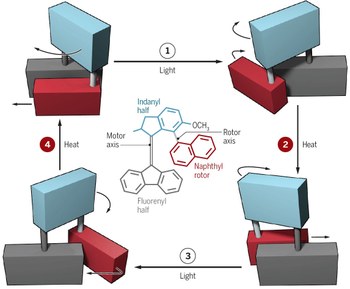
Gearing up molecular rotary motors
Massimo Baroncini, Alberto Credi
SCIENCE
Published online: 2 June 2017
2017, Vol. 356, Issue 6341, pp. 906-907
DOI: 10.1126/science.aan4353
Machines and motors based on synthetic small molecules are realized by a “bottom-up” approach to nanotechnology and could exploit molecular motion in one of two ways. The first generates macroscopic work by collecting the actions of many nanodevices organized in an array that provides spatial and temporal control of the motion activated by an energy supply. This approach mimics myosin motor proteins in skeletal muscles. The second route uses the energy-consuming directed movement of individual molecular machines to perform a task at the nanoscale, mimicking kinesin-based transport. Both cases mechanically couple an active component (the molecular machine) with nearby passive components and, ultimately, with the surrounding environment. Štacko et al. report the synchronous transmission of a photoactivated directional motion from a synthetic molecular motor to a coupled rotor. This demonstration takes an important step forward toward more complex mechanical functions with artificial nanoscale devices.